Text Solution
Verified by Experts
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
NCERT-COORDINATION COMPOUNDS-Exercise
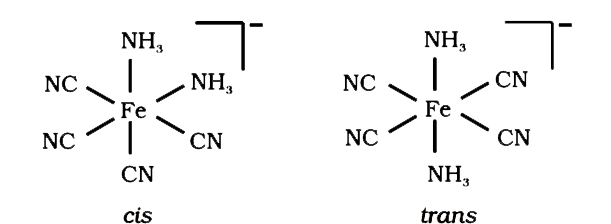
- Draw structures of geometrical isomers of [Fe(CN)(4)(NH(3))(2)]^(Θ) .
Text Solution
|
- Wrtie the formulas for the following coordination compounds: (i). Te...
Text Solution
|
- Write the IUPAC names of the following coordiantion compounds: (i) [...
Text Solution
|
- Indicate the type of isomerism exhibited by the following complexes an...
Text Solution
|
- Give evidence that [Co(NH3)5Cl]SO4 and [Co(NH3)5SO4]Cl are ionisation ...
Text Solution
|
- Deduce the structures of [NiCl(4)]^(2-) and [Ni (CN)(4)]^(2-) consider...
Text Solution
|
- [NiCl(4)]^(2-) is paramagnetic while [Ni(CO)(4)] is diamagnetic though...
Text Solution
|
- [Fe(H2O)6]^(3+) is strongly paramagnetic whereas [Fe(CN)6]^(3-) is wea...
Text Solution
|
- Explain [Co(NH3)6]^(3+) is an inner orbital complex whereas [Ni(NH3)6]...
Text Solution
|
- Predict the number of unpaired electrons in the square planar [Pt(CN)4...
Text Solution
|
- The hexaquo manganese(II) ion contains five unpaired electrons, while ...
Text Solution
|
- WERNER’S THEORY OF COORDINATION COMPOUNDS
Text Solution
|
- FeSO4 solution mixed with (NH4)2SO4 solution is 1:1 molar ratio gives ...
Text Solution
|
- Explain with two examples each of the following: coordination entity, ...
Text Solution
|
- What is meant by unidentate and ambidentate ligands? Give two examples...
Text Solution
|
- Specify the oxidation numbers of the metals in the following coordinat...
Text Solution
|
- Using IUPAC norms write the fomulas for the following (i). Tetrahydr...
Text Solution
|
- Using IUPAC norms write the systematic names of the following: (i). ...
Text Solution
|
- List various types of isomerism possible for coordination compounds, g...
Text Solution
|
- How many geometrical isomers are possible in the following corrdinatio...
Text Solution
|
- Draw the structures of optical isomers of: (i). [Cr(C2O4)3]^(3-) (...
Text Solution
|