A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
STATE OF MATTER (GASEOUS AND LIQUID STATE)
DINESH PUBLICATION|Exercise Selected straight objective types MCQs|58 VideosSTATE OF MATTER (GASEOUS AND LIQUID STATE)
DINESH PUBLICATION|Exercise Linked Comprhension type MCQs|11 VideosSTATE OF MATTER (GASEOUS AND LIQUID STATE)
DINESH PUBLICATION|Exercise Ulitmate Preparatory package|20 VideosS-BLOCK ELEMENTS (ALKALI AND ALKALINE EARTH METALS )
DINESH PUBLICATION|Exercise Assertion reason|12 VideosSTATES OF MATTER (SOLID STATE CHEMISTRY)
DINESH PUBLICATION|Exercise ULTIMATE PREPARATORY PACKAGE|21 Videos
Similar Questions
Explore conceptually related problems
DINESH PUBLICATION-STATE OF MATTER (GASEOUS AND LIQUID STATE)-Revision question from competitive exams
- The temperature of an ideal gas is increased from 27^(@)C to 127^(@)C,...
Text Solution
|
- By what factor does the average velocity of a gaseous molecule increas...
Text Solution
|
- 28 g each of the following gases are taken at 27^(@0C and 600 mm presu...
Text Solution
|
- a' and 'b' are van der Waals' constants for gases Chlorine is more eas...
Text Solution
|
- What will happen to volume of a bubble of air found under water in a l...
Text Solution
|
- The bubble of a gas released at the bottom of a lake increases to eig...
Text Solution
|
- Three different gases x, y, z of molecular masses 2, 16 and 64 were en...
Text Solution
|
- Choose the incorrect statement in the following
Text Solution
|
- 2 mole of N(2)O(4) (g) is kept in a closed container at 298 K and 1 at...
Text Solution
|
- 50 mL of each gas A and of gas B takes 150 and 200 seconds respectivel...
Text Solution
|
- For one mole of a van der Waals gas when b =0 and T =30 K the PV vs1//...
Text Solution
|
- A certain gas takes three times as long to effuse out as helium. Its m...
Text Solution
|
- For real gases, van der Waal's equation is written as [p + (an^(2))/(V...
Text Solution
|
- The compressibility factor for a real gas at high pressure is .
Text Solution
|
- Equal weight of CH(4) " and " H(2) are mixed in an empty container at ...
Text Solution
|
- A mixture contains 64 g of dioxygen and 60 g of neon at a total pressu...
Text Solution
|
- The gas with the highest critical temperature is
Text Solution
|
- Dipole-induced dipole interaction are present in which of the followin...
Text Solution
|
- For gaseous state, if most probable speed is denoted by C^(**) average...
Text Solution
|
- Maximum deviation from ideal gas is expected from
Text Solution
|
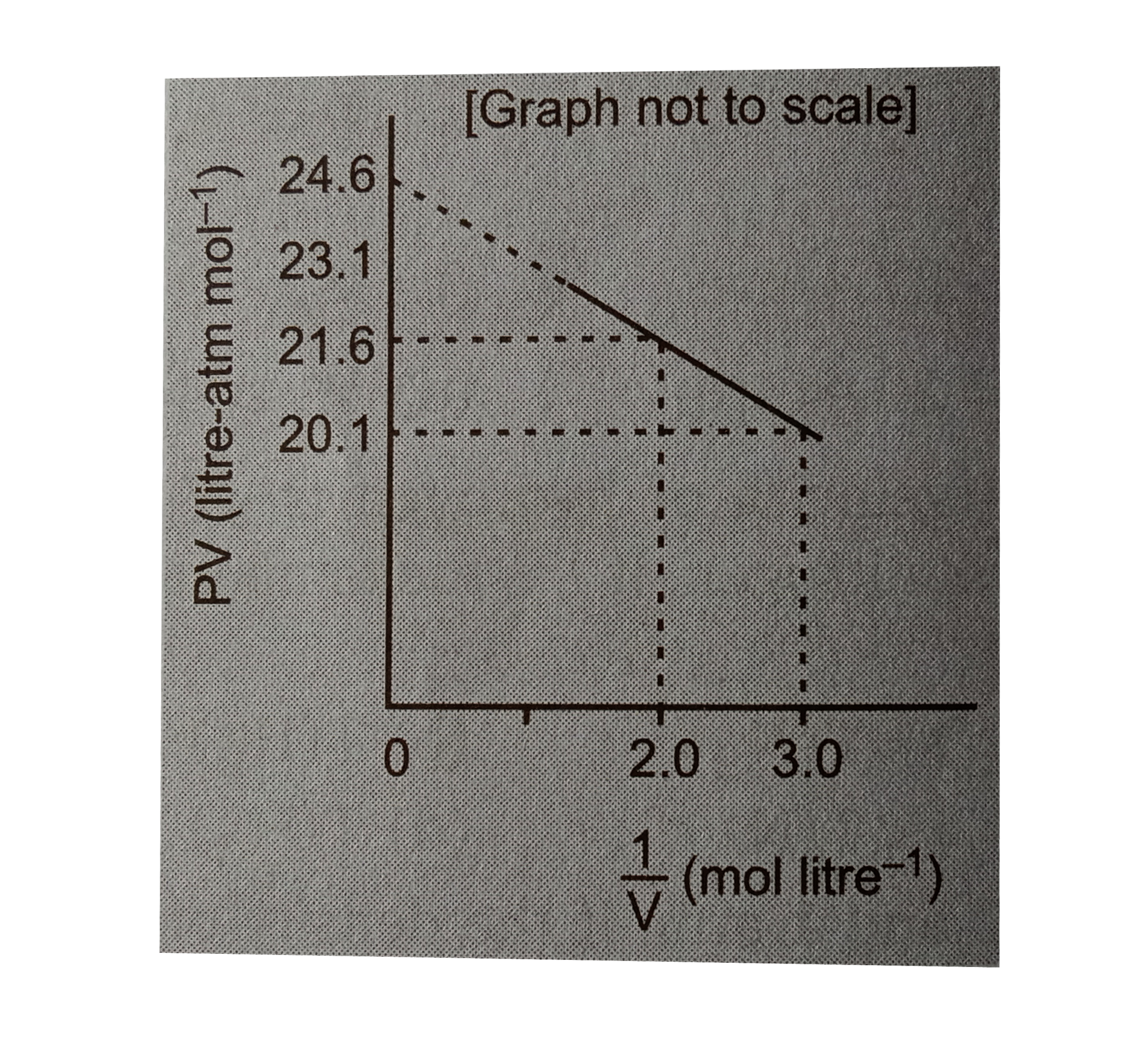 .
.