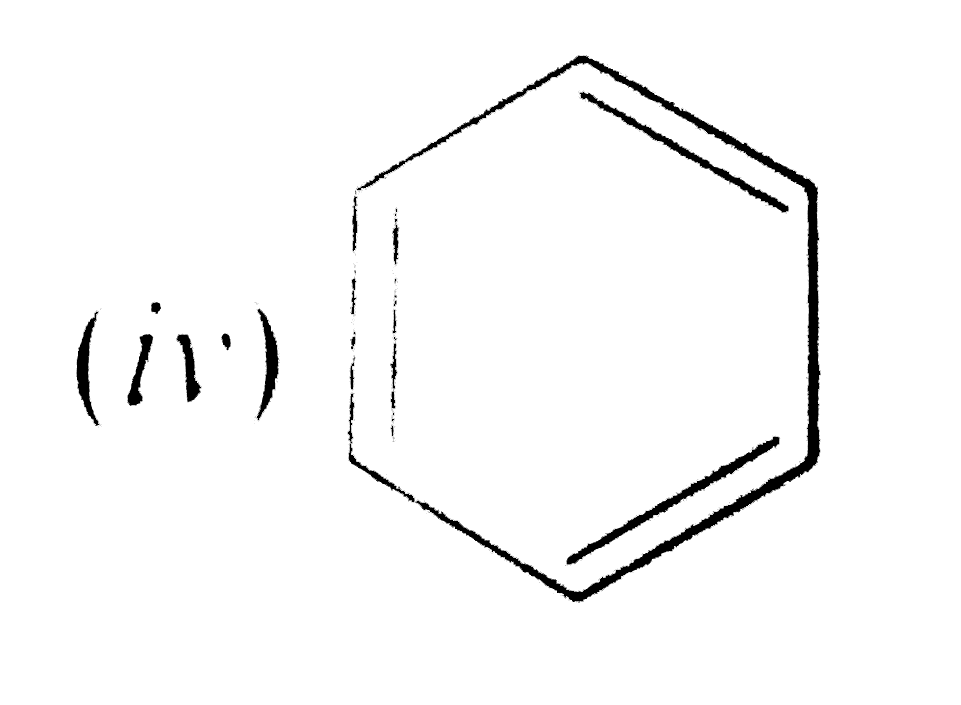
Text Solution
Verified by Experts
Topper's Solved these Questions
INTRODUCTION TO ORGANIC CHEMISTRY
OP TANDON|Exercise ILLUSTRATION OF OBJECTIVE QUESTIONS|12 VideosINTRODUCTION TO ORGANIC CHEMISTRY
OP TANDON|Exercise PROBLEMS FOR PRACTICE|12 VideosCHEMISTRY IN EVERYDAY LIFE
OP TANDON|Exercise SINGLE INTEGER ANSWER TYPE QUESTIONS|5 VideosMISCELLANEOUS (TOPICS OF GENERAL INTEREST)
OP TANDON|Exercise SET-VI: Problems on graphical aptitude|53 Videos
Similar Questions
Explore conceptually related problems
OP TANDON-INTRODUCTION TO ORGANIC CHEMISTRY -SINGLE INTEGER ANSWER TYPE
- Indicate the number of sigma and pi bonds in the following molecules: ...
Text Solution
|
- How many bonds (sigma and pi) are there in the following molecules? ...
Text Solution
|
- How many number of H-bonds are formed by a water molecule?
Text Solution
|
- How many resonating structures are possible for the compound furan
Text Solution
|
- The purine heterocycle occurs mainly in the structure of DNA. Identify...
Text Solution
|