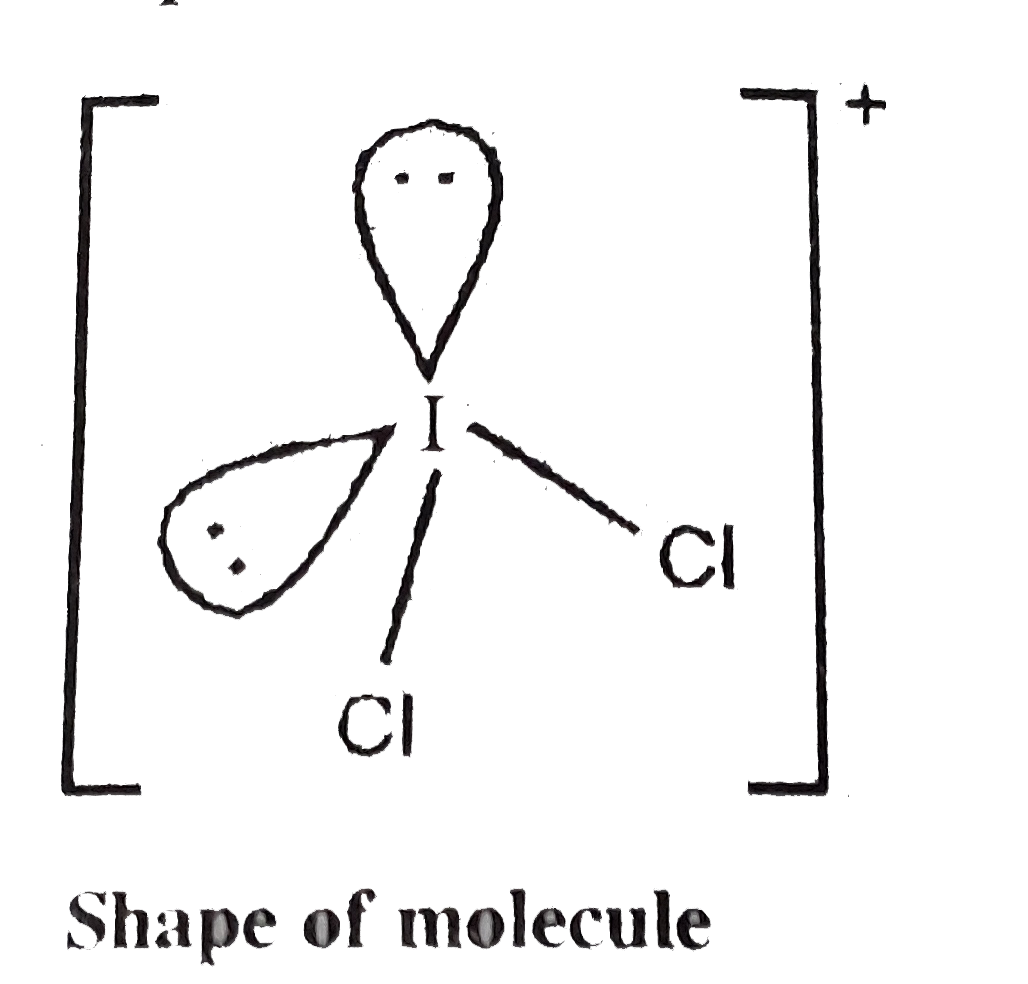
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
CHEMICAL BONDING AND MOLECULAR STRUCTURE
DINESH PUBLICATION|Exercise Brain Teaser-2|40 VideosCHEMICAL BONDING AND MOLECULAR STRUCTURE
DINESH PUBLICATION|Exercise Unit Test - 1|20 VideosCHEMICAL BONDING AND MOLECULAR STRUCTURE
DINESH PUBLICATION|Exercise Ultimate Preparatory Package|20 VideosCARBOXYLIC ACIDS
DINESH PUBLICATION|Exercise BRAIN STORMING MULTIPLE CHOICE QUESTIONS (MCQS)|13 VideosCHEMICAL KINETICS
DINESH PUBLICATION|Exercise Additional Numerical Problems For Practice|16 Videos
Similar Questions
Explore conceptually related problems
DINESH PUBLICATION-CHEMICAL BONDING AND MOLECULAR STRUCTURE-Brain Teaser-1
- Born-Haber cycle is used to determine
Text Solution
|
- Which of the following gaseous molecules is polar ?
Text Solution
|
- The incorrect order of decreasing boiling point is
Text Solution
|
- Which of the following statements is not regarding bonding molecular o...
Text Solution
|
- In the formation of N2^+ from N2, the electron is removed from
Text Solution
|
- The magnitude of lattice energy of a solid increases if
Text Solution
|
- The electronegativities of F, Cl, Br, and I are 4.0, 3.0, 2.8, and 2.5...
Text Solution
|
- In which of the following pairs does the first compound not have a hig...
Text Solution
|
- Which of the following compounds has the least tendency to form hydro...
Text Solution
|
- In which of the following set of molecules is the order of boiling poi...
Text Solution
|
- Which of the following statement is correct?
Text Solution
|
- Which among the following molecules is diamagnetic ?
Text Solution
|
- The energy of sigma(2s), is greater than that of sigma(1s)^** orbital ...
Text Solution
|
- A wedding ring presented to a bride contains 788 mg of gold and the re...
Text Solution
|
- The bond that stabilizes the secondary structure of proteins is
Text Solution
|
- Which of the following does not contain a coordinate bond?
Text Solution
|
- Hybridisation of central atom in IC l2^+ is
Text Solution
|
- The transition of an electron from a 4s orbital to ls orbital in hydro...
Text Solution
|
- The chemical species having same number of electrons in the outermost...
Text Solution
|
- From among the following triatomic species the least angle around the ...
Text Solution
|