Topper's Solved these Questions
CHEMICAL KINETICS
CBSE COMPLEMENTARY MATERIAL|Exercise LONG ANSWER TYPE QUESTIONS|12 VideosCHEMICAL KINETICS
CBSE COMPLEMENTARY MATERIAL|Exercise SHORT ANSWER-I TYPE QUESTIONS|26 VideosBIOMOLECULES
CBSE COMPLEMENTARY MATERIAL|Exercise SHORT ANSWER-II TYPE QUESTION|13 VideosCHEMISTRY IN EVERYDAY LIFE
CBSE COMPLEMENTARY MATERIAL|Exercise LONG ANSWER TYPE QUESTIONS|2 Videos
Similar Questions
Explore conceptually related problems
CBSE COMPLEMENTARY MATERIAL-CHEMICAL KINETICS-SHORT ANSWER TYPE-II QUESTIONS
- The activation energy of a reaction is 94.14 KJ/mol and the value of r...
Text Solution
|
- The rate constants of a reaction at 500 K and 700 K are 0.02s^(-1) and...
Text Solution
|
- The rate constants of a reaction at 700 K and 760 K are 0.011" M"^(-1)...
Text Solution
|
- The initial concentration of N2O5 in the first order reaction N2O5 to ...
Text Solution
|
- The following data were obtained during the first order thermal decomp...
Text Solution
|
- A first order reaction is 20% complete in 20 minutes. Calculate the ti...
Text Solution
|
- For a first order reaction, calculate the ratio between the time taken...
Text Solution
|
- The following results have been obtained during the kinetics studies o...
Text Solution
|
- The rate of reaction triples when the temperature changes from 293 K t...
Text Solution
|
- The decomposition of A into product has value of K as 4.5 xx 10^(3) se...
Text Solution
|
- Write rate law and order of the following reaction : AB+C2 to AB2C...
Text Solution
|
- Define activation energy of a reaction.
Text Solution
|
- What is the relation between rate constant and activation energy of a ...
Text Solution
|
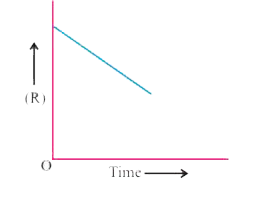
- For a chemical reaction R to P, the variation in the concentration (R)...
Text Solution
|
- For a chemical reaction R to P, the variation in the concentration (R)...
Text Solution
|
- For a chemical reaction R to P, the variation in the concentration (R)...
Text Solution
|
- What do you understand by a first order reaction ? Show that for a fir...
Text Solution
|
- In a pseudo first order reaction of hydrolysis of an ester in H2O, the...
Text Solution
|
- In a pseudo first order reaction of hydrolysis of an ester in H2O, the...
Text Solution
|
- The decomposition of NH3 on platinum surface is a zero order reaction....
Text Solution
|