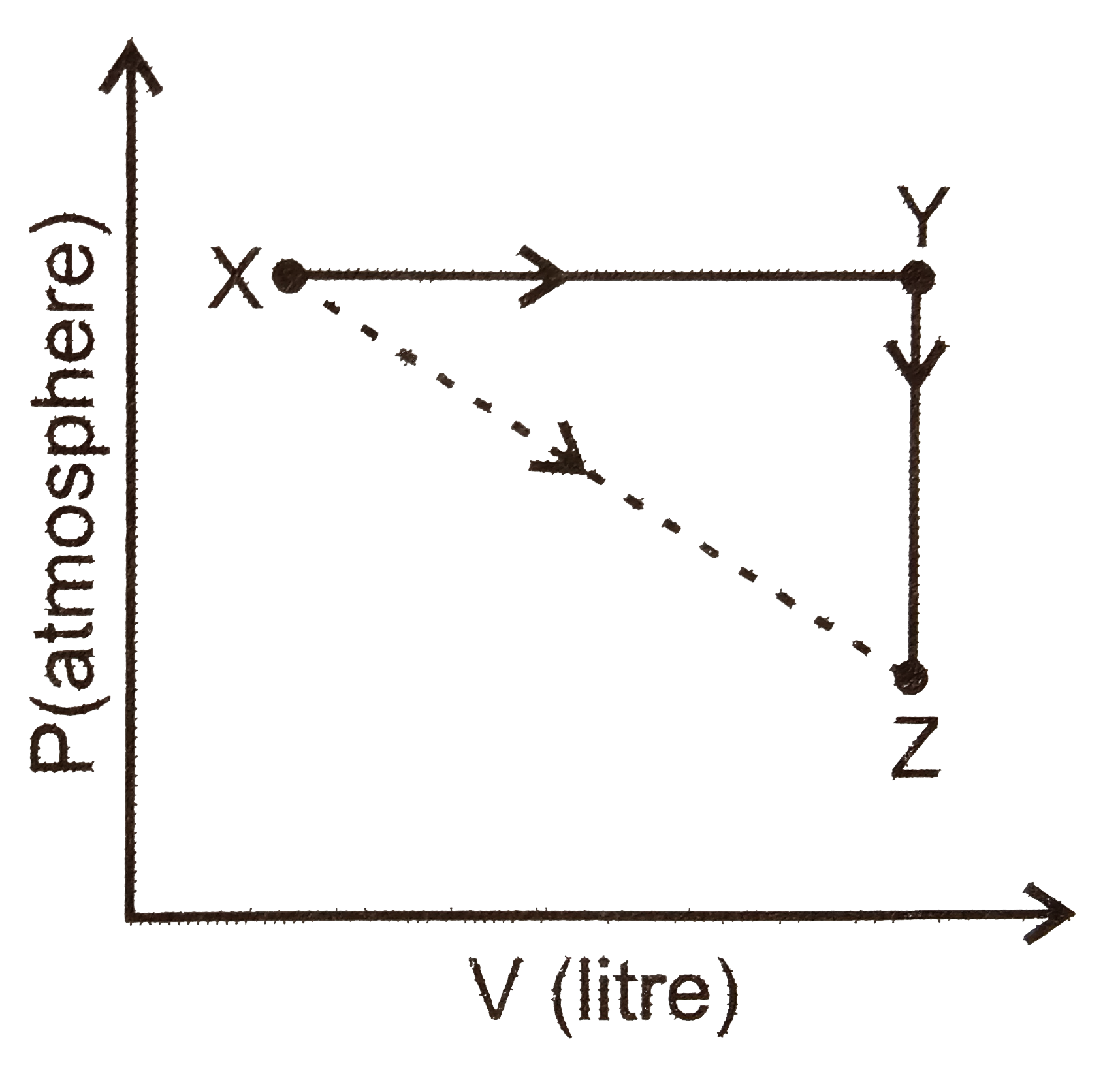
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
RESONANCE-THERMODYNAMICS-exercise-3 Part:(I)
- The direct conversion of A to B is difficult, hence is carried out by ...
Text Solution
|
- N(2) + 3H(2) hArr 2NH(3)" "K=4xx 10^(6)"at"298 " ...
Text Solution
|
- The value of log(10)K for a reaction A hArr B is (Given: Delta(f)H(298...
Text Solution
|
- For the process H(2)O(l) (1 "bar", 373 K) rarr H(2)O(g) (1"bar", 373 K...
Text Solution
|
- Statement -1: For every chmical reaction at equilibrium , standard Gid...
Text Solution
|
- Statement-1: There is a natural asymmetry between work to heat and con...
Text Solution
|
- Match the transformation in colums I with appropriate options in colum...
Text Solution
|
- For an ideal gas, consider only P-V work in going from an initial stat...
Text Solution
|
- For the process H(2)O(l)toH(2)O(g) at t=100^(@)C and 1 atmosphere pres...
Text Solution
|
- Match the thermodynamic processes given under column I with the expres...
Text Solution
|
- For a spontaneous reaction the Delta G, Equilibrium cosntant (K) and E...
Text Solution
|
- Identify the correct statement regarding a spontaneous process:
Text Solution
|
- In conversion of lime-stone to lime, CaCO(3(s)) to CaO((s)) + CO(2(g...
Text Solution
|
- Standard entropy of X(2) , Y(2) and XY(3) are 60, 40 and 50JK^(-1)mol...
Text Solution
|
- In a fuel cell methanol is used as fuel and oxygen gas is used as an o...
Text Solution
|
- For a particular reversible reaciton at temperature T, DeltaH and Delt...
Text Solution
|
- The entropy change involved in the isothermal reversible expansion of ...
Text Solution
|
- In view of the signs of Delta(r)G^(0) for the following reactions Pb...
Text Solution
|
- The incorrect expression among the following is
Text Solution
|
- The following reaction is performed at 298K 2NO(g) + O(2)(g) hArr 2...
Text Solution
|