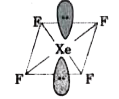
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
CHEMICAL BONDING AND MOLECULAR STRUCTURE
MODERN PUBLICATION|Exercise OBJECTIVE TYPE QUESTIONS (B. MULTIPLE CHOICE QUESTIONS)|40 VideosCHEMICAL BONDING AND MOLECULAR STRUCTURE
MODERN PUBLICATION|Exercise OBJECTIVE TYPE QUESTIONS (B. MULTIPLE CHOICE QUESTIONS) (JEE (MAIN) & OTHER STATE BOARDS FOR ENGINEERING ENTRANCE)|53 VideosCHEMICAL BONDING AND MOLECULAR STRUCTURE
MODERN PUBLICATION|Exercise HIGHER ORDER THINKING SKILLS|22 VideosCLASSIFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES
MODERN PUBLICATION|Exercise UNIT PRACTICE TEST|13 Videos
Similar Questions
Explore conceptually related problems
MODERN PUBLICATION-CHEMICAL BONDING AND MOLECULAR STRUCTURE-OBJECTIVE TYPE QUESTIONS (A. MULTIPLE CHOICE QUESTIONS)
- In an octahedral structure , the pair of d orbitals involved in d^(2)...
Text Solution
|
- Which of the following has highest bond angle?
Text Solution
|
- Which of the following molecules does not contain a lone pair of elect...
Text Solution
|
- In which of the following, the central atoms has two lone pairs of ele...
Text Solution
|
- What typs of hybridisation is possible in square planar molecules?
Text Solution
|
- PCl(5) molecule has the following geometry :
Text Solution
|
- In which of the following molecules are all the bonds not equal ?
Text Solution
|
- Which of the following molecules/ins does not contain unpaired electro...
Text Solution
|
- The bond order in O2^-ion is
Text Solution
|
- If molecular axis is Z then which of the following overlaping is not p...
Text Solution
|
- Which of the following is paramagnetic?
Text Solution
|
- Which one of the following pairs consists of only paramagnetic species
Text Solution
|
- The correct order of bond order values among the following (i) NO^(...
Text Solution
|
- Which of the following is not paramagnetic?
Text Solution
|
- The maximum bond strengths is in:
Text Solution
|
- which of the following hydrogen bond is strongest in vapour phase ?
Text Solution
|
- strongest hydrogen bonding is shown by
Text Solution
|
- Which of the following has highest boiling point?
Text Solution
|
- Which of the following has lowest boiling point?
Text Solution
|
- Which of the following statement is not true about amorphous solids?
Text Solution
|