Text Solution
Verified by Experts
Topper's Solved these Questions
CHEMICAL KINETICS
OSWAAL PUBLICATION|Exercise Topic - 2 (ORDER OF A REACTION , INTEGRATED RATE EQUATIONS AND HALF LIFE OF A REACTION )Long Answer Type Questions - II)|4 VideosCHEMICAL KINETICS
OSWAAL PUBLICATION|Exercise Topic - 2 (ORDER OF A REACTION , INTEGRATED RATE EQUATIONS AND HALF LIFE OF A REACTION ) (Short Answer Type Questions)|8 VideosBIOMOLECULES
OSWAAL PUBLICATION|Exercise TOPIC -II (LONG ANSWER TYPE QUESTIONS)|2 VideosCHEMISTY IN EVERYDAY LIFE
OSWAAL PUBLICATION|Exercise LONG ANSWER TYPE QUESTIONS-I|1 Videos
Similar Questions
Explore conceptually related problems
OSWAAL PUBLICATION-CHEMICAL KINETICS-Topic - 2 (ORDER OF A REACTION , INTEGRATED RATE EQUATIONS AND HALF LIFE OF A REACTION ) ( Long Answer Type Questions - I)
- Derive the integrated rate equation for rate constant of Zero order re...
Text Solution
|
- Derive an intergrated rate for the first order reaction.
Text Solution
|
- Thermal decomposition of a compound is of first order. If 50% of the c...
Text Solution
|
- Rate constant of a first order reaction A products is 0.016 min^(-1). ...
Text Solution
|
- The following data were obtained during the first order thermal decomp...
Text Solution
|
- Rate constant k for a first order reactions has been found to be 2.54 ...
Text Solution
|
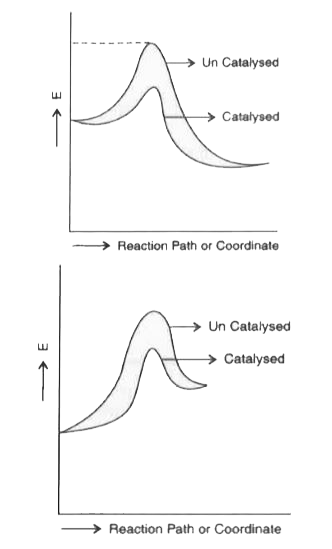
- With the help of a diagram, explain the physical significance of energ...
Text Solution
|
- The rate of a reaction becomes four times when the temperature changes...
Text Solution
|
- The rate of most reactions becomes double when their temperature is ra...
Text Solution
|
- Rate constant of reaction at 300 K and 400 K are 0.0345 S^(-1) and 0.1...
Text Solution
|
- The rate constant of a first order reaction at 300 K and 310 K are res...
Text Solution
|
- Define energy of activation. Draw a diagram of energy profile to show ...
Text Solution
|
- Half life of a first order reaction completes in 5 minutes. What perse...
Text Solution
|
- With graphical representation explain the effect of temperature on the...
Text Solution
|
- What will be the effect of temperature on rate constant ?
Text Solution
|
- The decomposition of A into products has a value of k as 4.5 xx 10^(3)...
Text Solution
|
- In general it is observed that the rate of a chemical reaction doubles...
Text Solution
|