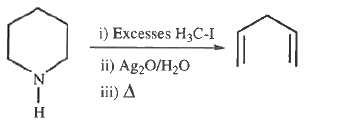A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
AMINES AND AZO COMPOUNDS
AAKASH SERIES|Exercise PRACTICE SHEET - 4 (More than one correct answer questions)|6 VideosAMINES AND AZO COMPOUNDS
AAKASH SERIES|Exercise PRACTICE SHEET - 4 (Linked Comprehension type questions) Passage - I|3 VideosAMINES AND AZO COMPOUNDS
AAKASH SERIES|Exercise PRACTICE SHEET - 3 (Integer answer type Questions)|7 VideosALKYL AND ARYL HALIDES
AAKASH SERIES|Exercise OBJECTIVE EXERCISE - 4 (ASSERTION (A) & REASON (R) TYPE QUESTIONS)|19 VideosAMINES AND DIAZONIUM SLATS
AAKASH SERIES|Exercise PRACTICE EXERCISE|25 Videos
Similar Questions
Explore conceptually related problems
AAKASH SERIES-AMINES AND AZO COMPOUNDS -PRACTICE SHEET - 4 (Single answer questions)
- The IUPAC name of the amine is :
Text Solution
|
- The pyramidal inversion from one invertomer of chiral tetrahedral amin...
Text Solution
|
- In the following reaction, CH(3)NH(2)+CHCl(3)+KOH to Nitrogen containi...
Text Solution
|
- Examine the following two structures for the anilinium ion and choose ...
Text Solution
|
- Complete the following reaction
Text Solution
|
- Complete the following reaction
Text Solution
|
- The product which is a red azo dye obtained on reacting benzene d...
Text Solution
|
- The best method for the preparation of primary amines is
Text Solution
|
- Acetophenone can be converted into amine is a single step by step
Text Solution
|
- The final product is
Text Solution
|
- N-ethyl formamide on treatment with POCI(3) in pyridine gives
Text Solution
|
- Piperidine is a secondary amine, which is subjected to Hofmann elimina...
Text Solution
|
- An organic compound with molecular formula C(3)H(5)N on hydrolysis giv...
Text Solution
|