Xenon Fluorides : Xenon reacts directly with fluorine on heating at 675 K in a sealed nickel tube. The product obtained depends upon the amount of fluorine present.
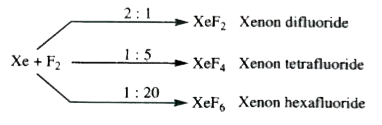
`XeF_2` reacts with `SbF_5` to form
Xenon Fluorides : Xenon reacts directly with fluorine on heating at 675 K in a sealed nickel tube. The product obtained depends upon the amount of fluorine present.
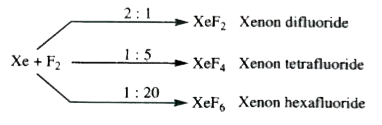
`XeF_2` reacts with `SbF_5` to form
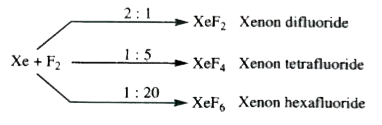
`XeF_2` reacts with `SbF_5` to form
A
`[XeF]^(+)[SbF_6]^(-)`
B
`[XeF_3]^(-) [ SbF_4]^(-)`
C
`Xe^(-)[PtF_6]^(+)`
D
`XeF_4`
Text Solution
Verified by Experts
The correct Answer is:
A
`XeF_2 + SbF_3 to [XeF]^(+) [ SbF_6]^(-)`
Topper's Solved these Questions
NOBLE GASES
AAKASH SERIES|Exercise LEVEL - II (LECTURE SHEET) (EXERCISE - III MATCH THE FOLLOWING QUESTIONS)|5 VideosNOBLE GASES
AAKASH SERIES|Exercise LEVEL - II (LECTURE SHEET) (EXERCISE - IV INTEGER ANSWER TYPE QUESTIONS)|10 VideosNOBLE GASES
AAKASH SERIES|Exercise LEVEL - II (LECTURE SHEET) (EXERCISE - I SIGGLE & ONE OR MORE THAN ONE CORRECT ANSWERS)|12 VideosNITROGEN CONTAINING COMPOUNDS
AAKASH SERIES|Exercise Conversions|18 VideosNUCLEAR CHEMISTRY
AAKASH SERIES|Exercise PRACTICE SHEET - 2 (Integer answer type Questions )|8 Videos
Similar Questions
Explore conceptually related problems
Xenon Fluorides : Xenon reacts directly with fluorine on heating at 675 K in a sealed nickel tube. The product obtained depends upon the amount of fluorine present. XeF_6 on complete hydrolysis gives
Xenon reacts with fluorine at 873 K and 7 bar to form XeF_(4) . In this reaction, the ratio of xenon and fluorine required is
K_2Cr_2O_7 acts as a good oxidizing agent in acidic medium underset("Orange")(Cr_(2)O_(7)^(2-)) + 14H^(+) + 6e^(-) rarr underset("Green")(2Cr^(3+)) + 7H_2O In alkaline solution, orange colour of Cr_(2)O_(7)^(2-) chages to yellow colour due to formation of Cr_2O_(4)^(2-) and again yellow colour changes to orange colour on changing the solution to acidic medium underset("Orange")(Cr_2O_7^(2-))+2OH^(_) rarrunderset("Yellow")(Cr_2O_7^(2-))+H_2O underset("Yellow")(2CrO_(4)^(2_-)) + 2H^(+) rarr underset("Orange")(Cr_(2)O_(7)^(2-) + H_(2)O) Cr_(4)^(2-) and Cr_(2)O_(7)^(2-) exist in equilibrium at pH =4 and are interconvertible by altering the pH of the solution. When heated with H_2SO_4 and metal chloride K_2Cr_2O_7 gives vapour of chromyl chloride (CrO_2Cl_2) . Chromyl chloride (CrO_2Cl_2) when passed into aqueous NaOH solution, yellow colour solution of CrO_(4)^(2-) is obtained. This on reaction with lead acetate gives yellow ppt. PbCrO_4 . When H_2O_2 is added to an acidified solution of dichromate ion, a complicated reaction occurs. The products obtained depend on the pH and concentration of dichromate. Cr_2O_7^(2-)+2H^(+) + 4H_(2)O_(2) rarr 2Cr(O_2)+5H_2O A deep blue-violet coloured peroxo compound, CrO(O_2)_2, ' called chromic peroxide is formed. This decomposes rapidly in aqueous solution into Cr^(3+) and xygen. CrO_3 on reaction with HCl and the product on reaction with NaOH(aq) give respectively
K_2Cr_2O_7 acts as a good oxidizing agent in acidic medium underset("Orange")(Cr_(2)O_(7)^(2-)) + 14H^(+) + 6e^(-) rarr underset("Green")(2Cr^(3+)) + 7H_2O In alkaline solution, orange colour of Cr_(2)O_(7)^(2-) chages to yellow colour due to formation of Cr_2O_(4)^(2-) and again yellow colour changes to orange colour on changing the solution to acidic medium underset("Orange")(Cr_2O_7^(2-))+2OH^(_) rarrunderset("Yellow")(Cr_2O_7^(2-))+H_2O underset("Yellow")(2CrO_(4)^(2_-)) + 2H^(+) rarr underset("Orange")(Cr_(2)O_(7)^(2-) + H_(2)O) Cr_(4)^(2-) and Cr_(2)O_(7)^(2-) exist in equilibrium at pH =4 and are interconvertible by altering the pH of the solution. When heated with H_2SO_4 and metal chloride K_2Cr_2O_7 gives vapour of chromyl chloride (CrO_2Cl_2) . Chromyl chloride (CrO_2Cl_2) when passed into aqueous NaOH solution, yellow colour solution of CrO_(4)^(2-) is obtained. This on reaction with lead acetate gives yellow ppt. PbCrO_4 . When H_2O_2 is added to an acidified solution of dichromate ion, a complicated reaction occurs. The products obtained depend on the pH and concentration of dichromate. Cr_2O_7^(2-)+2H^(+) + 4H_(2)O_(2) rarr 2Cr(O_2)+5H_2O A deep blue-violet coloured peroxo compound, CrO(O_2)_2, ' called chromic peroxide is formed. This decomposes rapidly in aqueous solution into Cr^(3+) and xygen. Which of the following statements is wrong when a mixture of NaCI and K_2Cr_2O_2 is gently walmed with conc. H_2SO_4 ?
K_2Cr_2O_7 acts as a good oxidizing agent in acidic medium underset("Orange")(Cr_(2)O_(7)^(2-)) + 14H^(+) + 6e^(-) rarr underset("Green")(2Cr^(3+)) + 7H_2O In alkaline solution, orange colour of Cr_(2)O_(7)^(2-) chages to yellow colour due to formation of Cr_2O_(4)^(2-) and again yellow colour changes to orange colour on changing the solution to acidic medium underset("Orange")(Cr_2O_7^(2-))+2OH^(_) rarrunderset("Yellow")(Cr_2O_7^(2-))+H_2O underset("Yellow")(2CrO_(4)^(2_-)) + 2H^(+) rarr underset("Orange")(Cr_(2)O_(7)^(2-) + H_(2)O) Cr_(4)^(2-) and Cr_(2)O_(7)^(2-) exist in equilibrium at pH =4 and are interconvertible by altering the pH of the solution. When heated with H_2SO_4 and metal chloride K_2Cr_2O_7 gives vapour of chromyl chloride (CrO_2Cl_2) . Chromyl chloride (CrO_2Cl_2) when passed into aqueous NaOH solution, yellow colour solution of CrO_(4)^(2-) is obtained. This on reaction with lead acetate gives yellow ppt. PbCrO_4 . When H_2O_2 is added to an acidified solution of dichromate ion, a complicated reaction occurs. The products obtained depend on the pH and concentration of dichromate. Cr_2O_7^(2-)+2H^(+) + 4H_(2)O_(2) rarr 2Cr(O_2)+5H_2O A deep blue-violet coloured peroxo compound, CrO(O_2)_2, ' called chromic peroxide is formed. This decomposes rapidly in aqueous solution into Cr^(3+) and xygen. What happens when a solution of potassium chromate is treated with an excess of dilute nitric acid?
One mole of fluorine reacted with two moles of hot concentrated KOH. The products formed are KF, H_(2)O and O_(2) . Themolar ratio of KF, H_(2)O and O_(2) respectively is
Chemical reactions are invariably associated with the transfer of energy either in the form of hear or light. In the laboratory, heat changes in physical and chemical processes are measured with an instrument called calorimeter. Heat change in the process is calculated as: q= ms Delta T , s= Specific heat = c Delta T = Heat capacity. Heat of reaction at constant pressure is measured using simple or water calorimeter. Q_(v)= Delta U = Internal energy change, Q_(P) = DeltaH, Q_(P) = Q_(V) + P Delta V and DeltaH = Delta U+ Delta nRT . The amount of energy released during a chemical change depends on the physical state of reactants and products, the condition of pressure, temperature and volume at which the reaction is carried out. The variation of heat of reaction with temperature and pressure is given by Kirchoff's equation: (DeltaH_(2) - DeltaH_(1))/(T_(2)-T_(1))= Delta C_(P) (At constant pressure), (DeltaU_(2) - DeltaU_(1))/(T_(2)-T_(1)) = DeltaC_(V) (At constant volume) The enthalpy change (DeltaH) for the reaction N_(2) (g) + 3H_(2)(g) rarr 2NH_(3)(g) is -92.38kJ at 298 K. The internal energy change DeltaU at 298 K is
How many of the following are correct statements. -Valency of an element is always a whole number. - In neutrilisation reaction between sulphuric acid and potassium SO_(4)^(--) and K^(+) ion are spectator ions. -Leade is ………..stable in +2 oxidation state due to inert pair effect. -Thallium salts in +3 oxidation state act as good reducing agents. -2H_(2)S+SO_(2)to3S+2H_(2)O is disproportionation reacts. -Fluorine cannot be obtained from fluoride by chemical methods.
On the basis of the postulates of kinetic theory of gases, it is possible to derive the mathematical expression, commonly known as kinetic gas equation. PV = 1/3 m n u^3? where, P= Pressure of the gas, V a volume of the gas, m=Mass of a molecule, n = Number of molecules present in the given amount of a gas and u = root mean square speed For one mole of gas, PV = RT and n=N_A 1/3 m N_a u^2 = RT or 2/3 .1/2m N_A u^2 = N_A [1/2mN_Au^2 = KE "per mole"] ,2/3K.E. = RT implies K.E. 3/2RT Average kinetic energy per mol does not depend on the nature of the gas but depends only on temperature. This, when two gases are mixed at the same temperature, there will be no rise or decrease in temperature unless both react chemically. Average kinetic energy per molecule = ("Average K.E. per mole")/N = 3/2(RT)/(N) implies 3/2kT where k is the Boltzmann constant Which of the following expressions correctly represents the relationship between the average molar kinetic energies of CO and N_2 molecules at the same temperature ?
AAKASH SERIES-NOBLE GASES-LEVEL - II (LECTURE SHEET) (EXERCISE - II LINKED COMPREHENSION TYPE QUESTIONS)
- Noble gases A, B, C, D, E are passed through Dewar's flask at-100^@C A...
Text Solution
|
- Noble gases A, B, C, D, E are passed through Dewar's flask at-100^@C A...
Text Solution
|
- Xenon Fluorides : Xenon reacts directly with fluorine on heating at 67...
Text Solution
|
- Xenon Fluorides : Xenon reacts directly with fluorine on heating at 67...
Text Solution
|