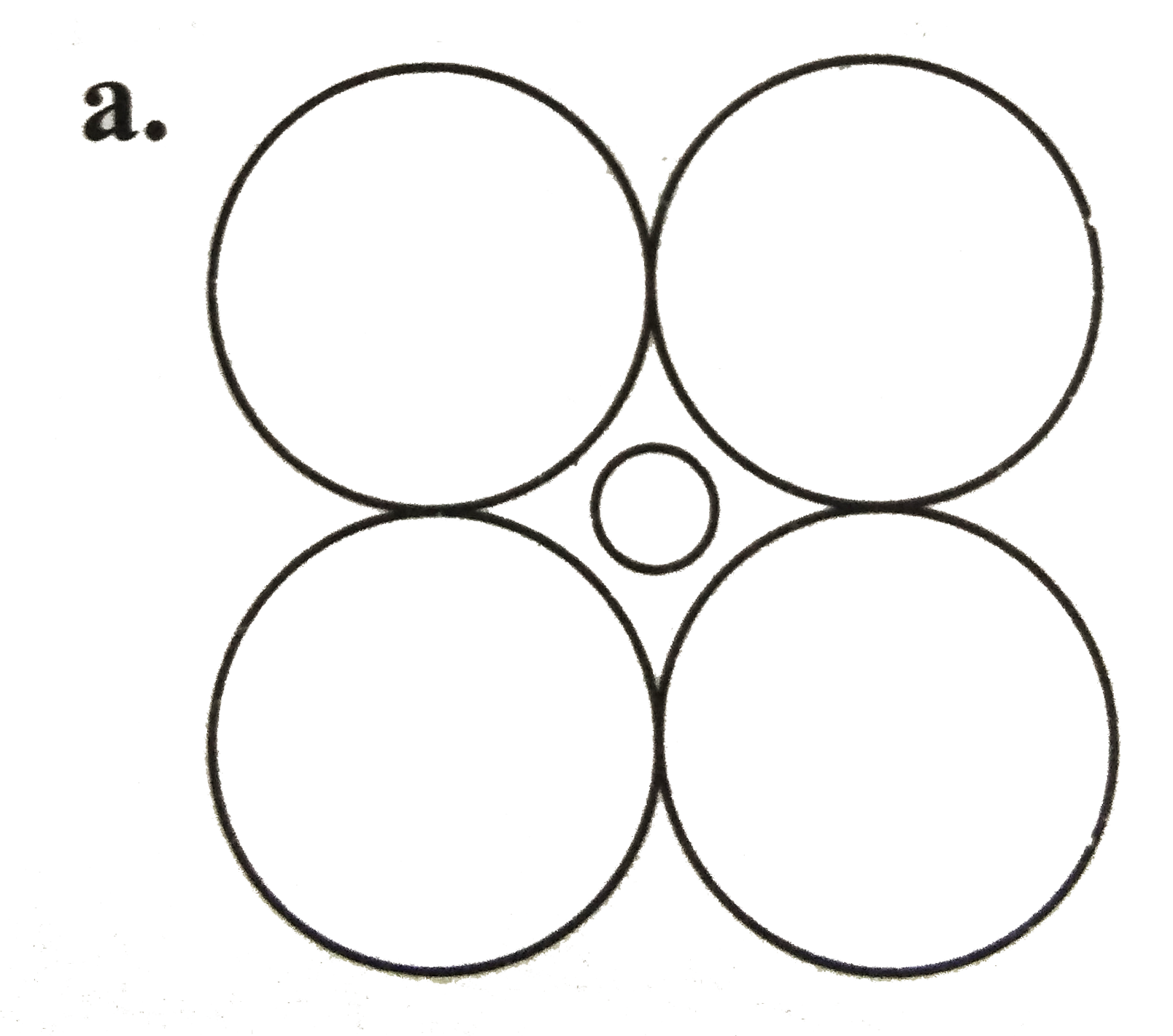
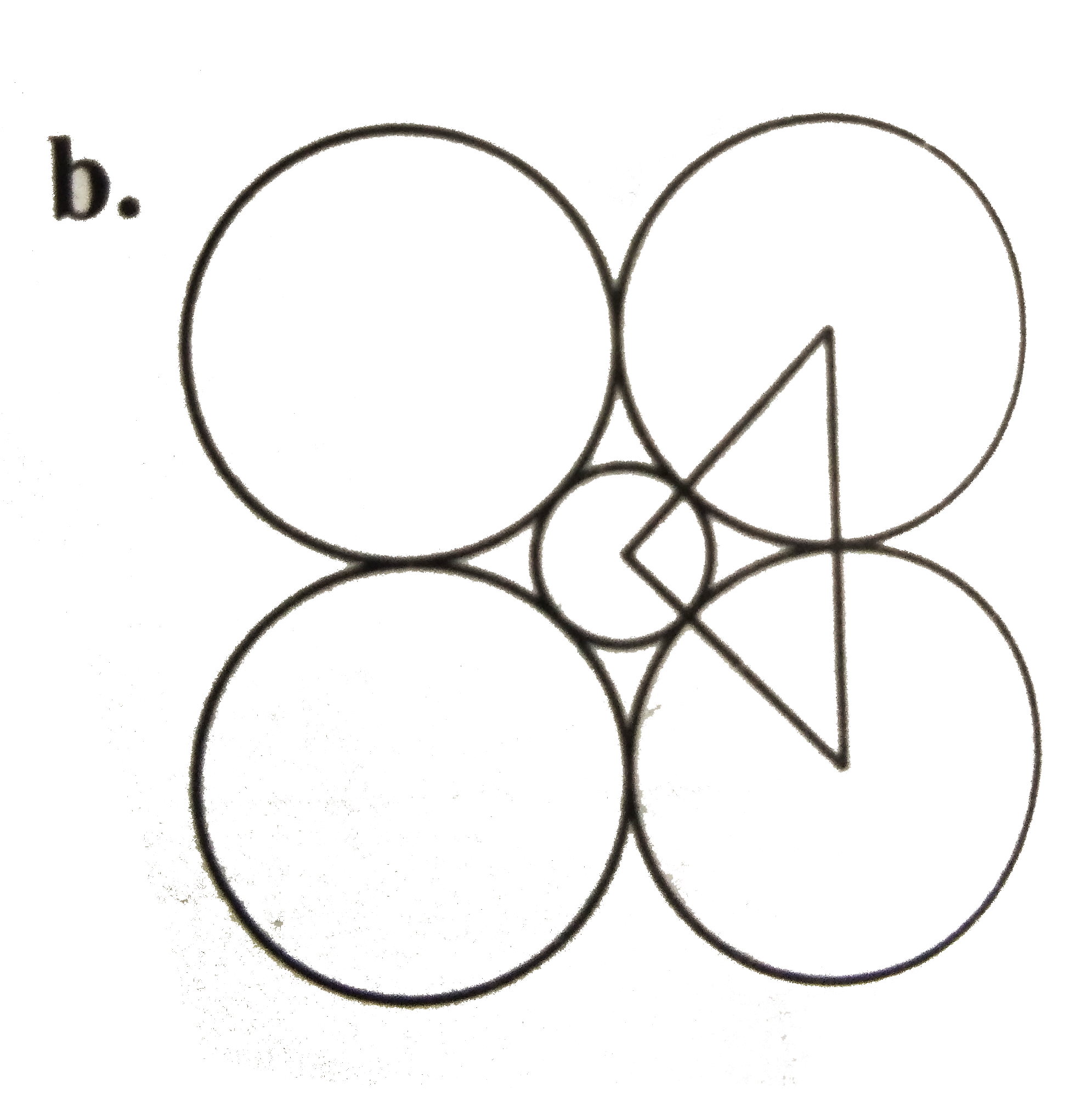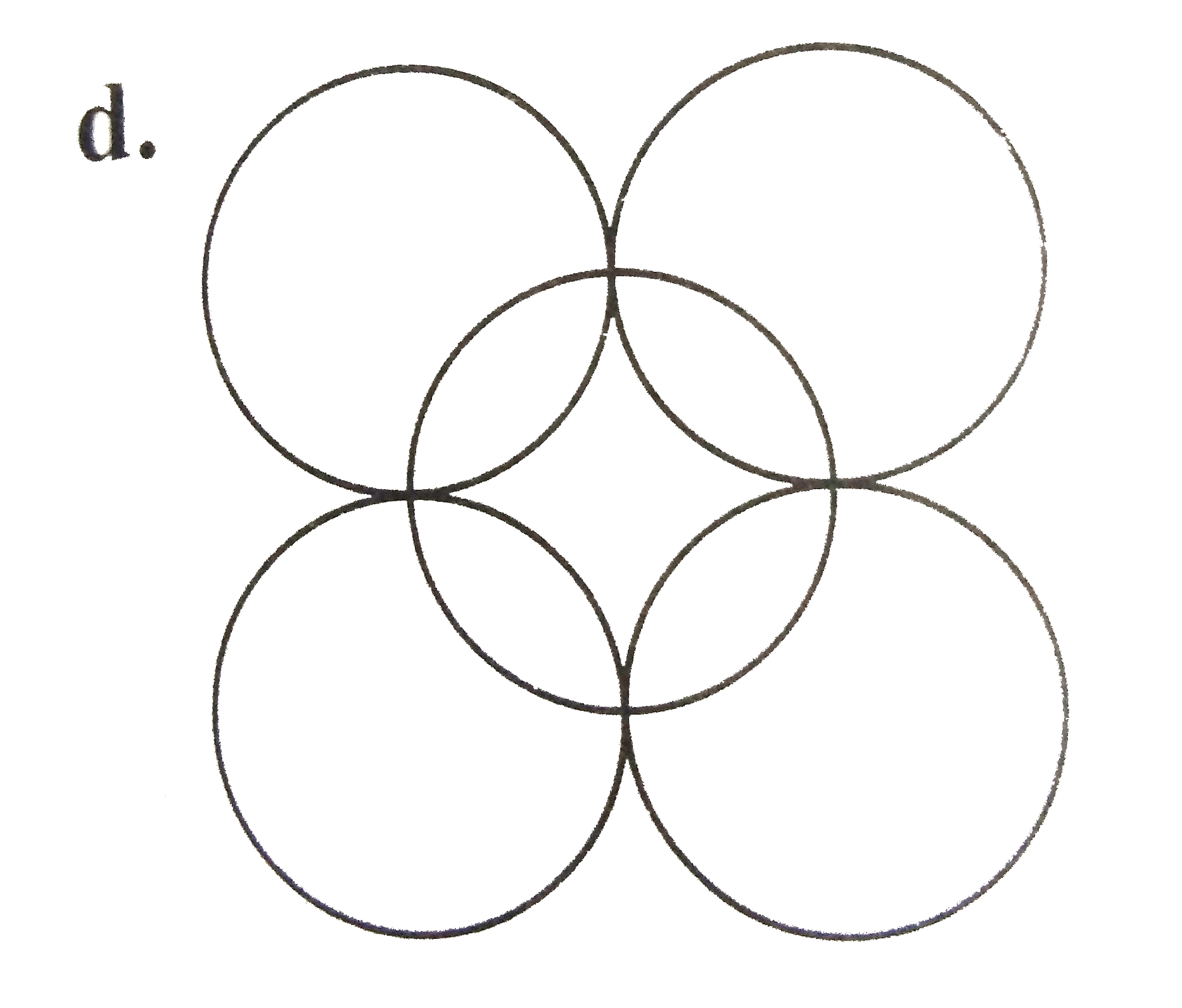A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
SOLID STATE
CENGAGE CHEMISTRY ENGLISH|Exercise Exercises (Multiple Correct)|39 VideosSOLID STATE
CENGAGE CHEMISTRY ENGLISH|Exercise Exercises (Single Correct)|72 VideosSOLID STATE
CENGAGE CHEMISTRY ENGLISH|Exercise Solved Examples|13 VideosREDUCTION AND OXIDATION REACTION OF ORGANIC COMPOUNDS
CENGAGE CHEMISTRY ENGLISH|Exercise SUBJECTIVE TYPE|4 VideosSOLUTIONS
CENGAGE CHEMISTRY ENGLISH|Exercise Ex 2.3 (Objective)|9 Videos
Similar Questions
Explore conceptually related problems
CENGAGE CHEMISTRY ENGLISH-SOLID STATE-Exercises (Linked Comprehension)
- If calcium crystallizes in bc c arrangement and the radius of Ca atom ...
Text Solution
|
- The number of unit cells in the Ca atom lies on the surface of a cubic...
Text Solution
|
- The number of unit cells in the Ca atom lies on the surface of a cubic...
Text Solution
|
- The "OLIVINE" series of minerals consists of crystal in which Fe^(2+) ...
Text Solution
|
- The "OLIVINE" series of minerals consists of crystal in which Fe^(2+) ...
Text Solution
|
- AX,AY,BX, and BY have rock salt type structure with following internuc...
Text Solution
|
- AX,AY,BX, and BY have rock salt type structure with following internuc...
Text Solution
|
- AX,AY,BX, and BY have rock salt type structure with following internuc...
Text Solution
|
- AX,AY,BX, and BY have rock salt type structure with following internuc...
Text Solution
|
- AX,AY,BX, and BY have rock salt type structure with following internuc...
Text Solution
|
- In a unit cell, atoms (A) are present at all corner lattices, (B) are ...
Text Solution
|
- In a unit cell, atoms (A) are present at all corner lattices, (B) are ...
Text Solution
|
- In a unit cell, atoms (A) are present at all corner lattices, (B) are ...
Text Solution
|