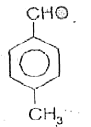
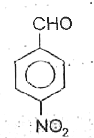
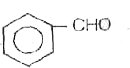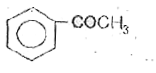A
B
C
D
Text Solution
AI Generated Solution
The correct Answer is:
Similar Questions
Explore conceptually related problems
Recommended Questions
- Which one is most reactive towards nucleophilic addition reaction?
Text Solution
|
- Which one is most reactive towards nucleophilic addition reaction?
Text Solution
|
- Which of the following compounds is most reactive towards nucleophilic...
Text Solution
|
- Which one is most reactive towards nucleophilic addition reaction?
Text Solution
|
- Which of the following compounds is most reactive towards nucleophilic...
Text Solution
|
- Which of the following compounds is most reactive towards nucleophilic...
Text Solution
|
- Which one is most reactive towards nucleophilic addition reaction?
Text Solution
|
- Which one is most reactive towards Nucleophilic addition reaction
Text Solution
|
- Which of the following compounds is most reactive towards nucleophilic...
Text Solution
|