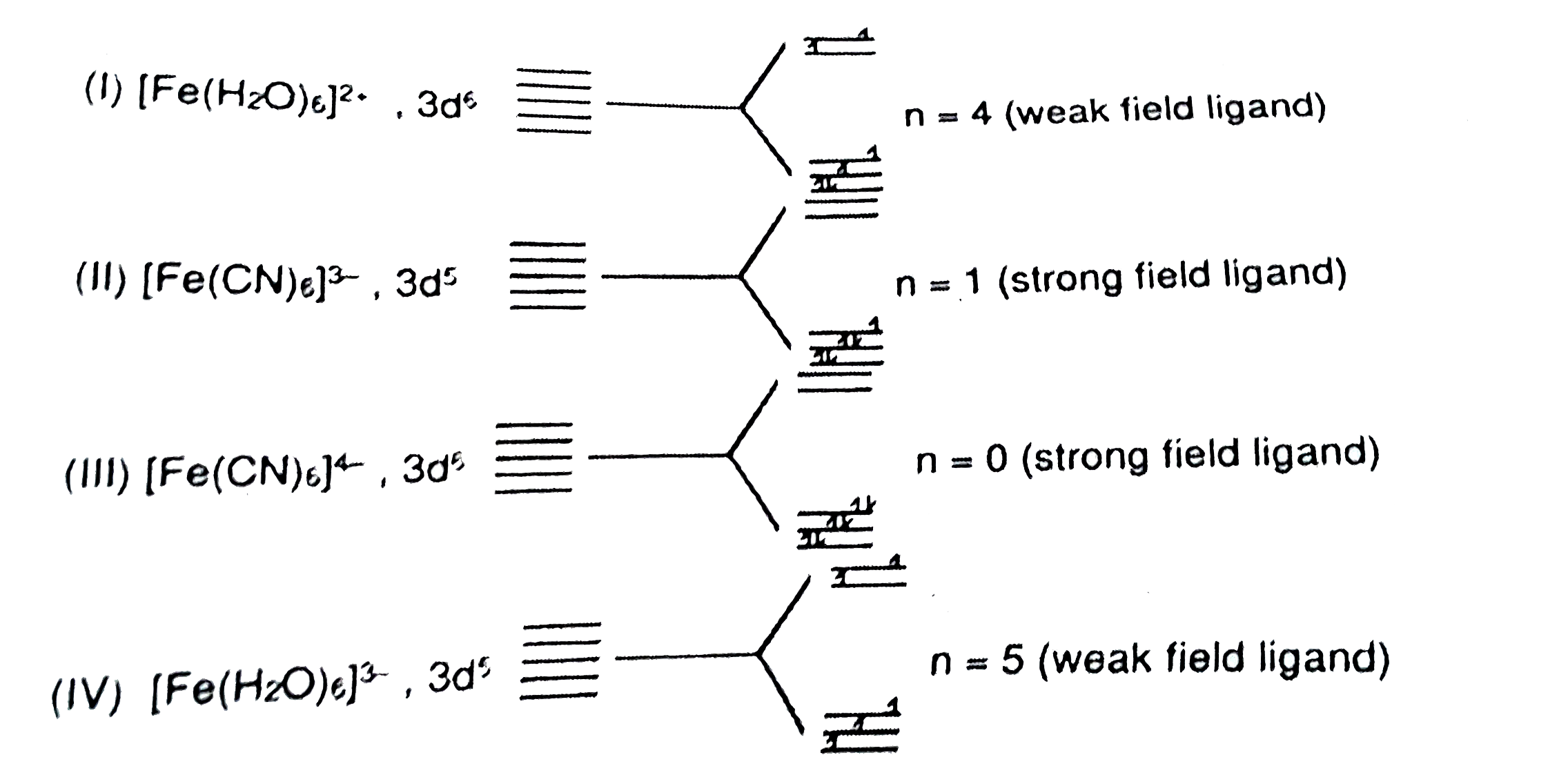
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
COORDINATION COMPOUNDS
RESONANCE ENGLISH|Exercise Additional Problem for Self Practice (APSP) Part-II|61 VideosCOORDINATION COMPOUNDS
RESONANCE ENGLISH|Exercise Additional Problem for Self Practice (APSP) Part-III Subjected Question|7 VideosCOORDINATION COMPOUNDS
RESONANCE ENGLISH|Exercise Exercise-3 Online Exam|22 VideosCHEMISTRY IN EVERYDAY LIFE
RESONANCE ENGLISH|Exercise ORGANIC CHEMISTRY(Chemistry in every day life)|31 VideosD & F BLOCK ELEMENTS
RESONANCE ENGLISH|Exercise ORGANIC CHEMISTRY(Aldehydes , Ketones, Carboxylic acid)|15 Videos
Similar Questions
Explore conceptually related problems
RESONANCE ENGLISH-COORDINATION COMPOUNDS-Additional Problem for Self Practice (APSP) Part-I
- If excess of AgNO(3) solution is added to 100 mL of a 0.024 M solution...
Text Solution
|
- A complex of a certain metal ion has a magnetic moment of 4.90BM Anoth...
Text Solution
|
- What will be oxidation state iron in given complex (Product)? Na(2)[...
Text Solution
|
- Aqueous solution of nickel sulphate on treating with pyridine and then...
Text Solution
|
- In the compound, lithium tetrahydroaluminate, the ligand is
Text Solution
|
- The oxidation number of Co in the complex ion
Text Solution
|
- The magnitude of crystal field stabilisation energy (CFSE of Delta(1))...
Text Solution
|
- Other than the X-ray difference , how could be the following pairs of ...
Text Solution
|
- [Fe(e n)(2)(H(2)O)(2)]^(2+) +e n to complex (x). The correct statement...
Text Solution
|
- Which of the following pairs will show the same magnetic moment ('spin...
Text Solution
|
- What will be the 'spin only' magnetic moment of the complex formed whe...
Text Solution
|
- Which of the following statement about Fe(CO)(5) is correct ?
Text Solution
|
- The crystal field -splitting for Cr^(3+) ion in octahedral field chang...
Text Solution
|
- Which of the following complex ions is not expected to absorb visible ...
Text Solution
|
- Of the following complex ions, the one that probably has the largest o...
Text Solution
|
- The correct structure of Fe(CO)(5) is (Z = 26 for Fe)
Text Solution
|
- Arrange the following in order of decreasing number of unpaired electr...
Text Solution
|
- Match List-I (Complexes)with List-II (Hybridization ) of central atom ...
Text Solution
|
- Which of the following complexes ionization isomerism ?
Text Solution
|
- Coordination number of Ni in [(C2O4)3]^(–4) is
Text Solution
|