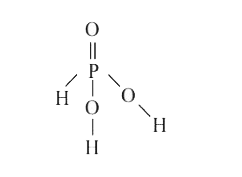
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
VMC MODULES ENGLISH-STOICHIOMETRY - I-JEE Advanced (Archive)
- Three grams of salt of molecular weight 30 is dissolved in 250 g of wa...
Text Solution
|
- If 0.5 mole of BaCl(2) mixed with 0.20 mole of Na(3) PO(4) the maximum...
Text Solution
|
- The density of a 3 M Na(2) S(2) O(3) (sodium thiosulphate) solution is...
Text Solution
|
- A molal solution is one that contains 1 mol of a solute in
Text Solution
|
- In which mode of expression, the concentration of a solution remains i...
Text Solution
|
- A sugar syrup of weight 214.2 g contains 34.2 g of sugar (C(12)H(2...
Text Solution
|
- Calculate the molality of 1 litre solution of 93% H2SO4 (weight/volume...
Text Solution
|
- A solid mixture(5.000 g) consisting of lead nitrate and sodium nitrate...
Text Solution
|
- The weight of 1 xx 10^22 molecules of CuSO4. 5H2O is:
Text Solution
|
- 8.0575 xx 10^(-12) kg of Glauber's salt are dissolved in water to obta...
Text Solution
|
- A plant virus is found to consist of uniform cylindrical particle of 1...
Text Solution
|
- The normality of 0.3 M phosphorous acid (H(3) PO(3)) is
Text Solution
|
- How many moles of electrons weigh one kilogram?
Text Solution
|
- An aqueous solution of 6.3 gm oxalic acid dihydrate is made upto Th...
Text Solution
|
- Which has maximum number of atoms:
Text Solution
|
- Calculate the molarity of water if its density is 1000 kg m^(-3)
Text Solution
|
- Dissolving 120g of urea (Mw = 60) in 1000 g of water gave a solution o...
Text Solution
|
- 29.2% (w//w) HCl stock, solution has a density of 1.25 g mL^(-1). The ...
Text Solution
|
- A compound H(2)X with molar weigth of 80g is dissolved in a solvent ha...
Text Solution
|
- The mole fraction of a solute in a solutions is 0.1. At 298K molarity ...
Text Solution
|