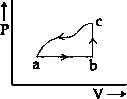
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
GASEOUS STATE & THERMODYNAMICS
VMC MODULES ENGLISH|Exercise JEE ADVANCED (ARCHIVE )|111 VideosGASEOUS STATE & THERMODYNAMICS
VMC MODULES ENGLISH|Exercise Level - 2|40 VideosENERGY & MOMENTUM
VMC MODULES ENGLISH|Exercise JEE ADVANCE (ARCHIVE) - TRUE/FALSE TYPE|1 VideosGRAVITATION
VMC MODULES ENGLISH|Exercise JEE Advance (Archive) TRUE/FALSE|1 Videos
Similar Questions
Explore conceptually related problems
VMC MODULES ENGLISH-GASEOUS STATE & THERMODYNAMICS-JEE MAIN (ARCHIVE )
- An ideal gas is enclosed in a cylinder at pressure of 2atm and tempera...
Text Solution
|
- An ideal gas occupies a volume of 2m^(3) at a pressure of 3xx10^(6...
Text Solution
|
- for the given cyclic process CAB as shown for a gas , the work don...
Text Solution
|
- The pressure exerted by the gas molecule is
Text Solution
|
- The given diagrams shows four processes i.e., isochoric , isobaric , i...
Text Solution
|
- The temperature ,a t which the root mean square velcity of hydrogen mo...
Text Solution
|
- For a given gas at 1 atm pressure, rms speed of the molecules is 200 m...
Text Solution
|
- An HCl molecule has rolational, translational and vibrational motions....
Text Solution
|
- Figure shows two processes a and b for a given sample of a gas. If Del...
Text Solution
|
- The specific heats, C(P) and C(V) of a gas of diatomic molecules, A ar...
Text Solution
|
- When heat Q is supplied to a diatomic gas of rigid molecules, at const...
Text Solution
|
- One mole of an ideal gas passes through a process where pressure and v...
Text Solution
|
- A cylinder with fixed capacity of 67.2 lit contains helium gas at STP....
Text Solution
|
- A 25xx10^(-3)m^(3) volume cylinder is filled with 1 mol of O2 gas at r...
Text Solution
|
- n moles of an ideal gas with constant volume heat capacity CV undergo ...
Text Solution
|
- The efficiency of a heat engine is 1//6Its efficiency double when the ...
Text Solution
|
- The number density of molecules of a gas depends on their distance r f...
Text Solution
|
- A diatomic gas with rigid molecules does 10J of work when expanded at...
Text Solution
|
- Two moles of helium gas is mixed with three moles of hydrogen molecule...
Text Solution
|
- A sample of an ideal gas is taken through the cyclic process abca and ...
Text Solution
|