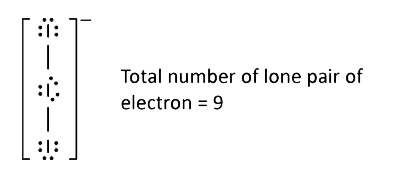A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
CHEMICAL BONDING-I & II
VMC MODULES ENGLISH|Exercise JEE Advanced (Archive)|98 VideosCHEMICAL BONDING-I & II
VMC MODULES ENGLISH|Exercise JEE Main (Numerical Value Questions)|15 VideosCHEMICAL BONDING & MOLECULAR STRUCTURE
VMC MODULES ENGLISH|Exercise IN-CHAPTER EXERCISE-L|9 VideosCHEMICAL EQUILIBRIUM
VMC MODULES ENGLISH|Exercise IN-CHAPTER EXERCISE - G|10 Videos
Similar Questions
Explore conceptually related problems
VMC MODULES ENGLISH-CHEMICAL BONDING-I & II-JEE Main (Archive)
- Which of the following species is not paramagnetic?
Text Solution
|
- The group having isoelectronic species is
Text Solution
|
- Which of the following is paramagnetic ?
Text Solution
|
- The group having triangular planar structures is :
Text Solution
|
- Total number of lone pair of electrons in I(3)^(-) ion is
Text Solution
|
- Which of the following compounds contain(s) no covalent bond(s)? K...
Text Solution
|
- According to molecular orbital theory, which of the following will not...
Text Solution
|
- During the change of O(2) to O(2)^(-), the incoming electron goes to t...
Text Solution
|
- The oxoacid of sulphur that does not contain bond between sulphur atom...
Text Solution
|
- Among the following, the molecule expected to be stabilized by anion f...
Text Solution
|
- The basic structural unit of feldspar, zeolites, mica, and asbestos is...
Text Solution
|
- The correct statement among the following is:
Text Solution
|
- Among the following species, the diamagnetic molecule is :
Text Solution
|
- The ion that has sp ^ 3 d ^ 2 hybridization for the central ...
Text Solution
|
- Among the following molecules/ions C(2)^(2-),N(2)^(2-),O(2)^(2-),O(2) ...
Text Solution
|
- The correct statement about ICl(5) and ICl(4)^(-) is
Text Solution
|
- According to molecular orbital theory, which of the following is true ...
Text Solution
|
- In which of the following processes, the bond order has increased and ...
Text Solution
|
- Two pi and half sigma bonds are present in:
Text Solution
|
- The type of hybridisation and number of lone pair(s) of electrons of X...
Text Solution
|