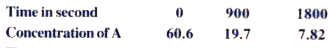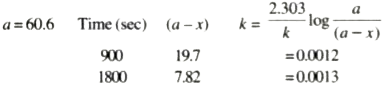Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
CHEMICAL KINETICS
ICSE|Exercise INTEXT QUESRTIONS|81 VideosCHEMICAL KINETICS
ICSE|Exercise EXERCISE (PART- I (OBJECTIVE QUESRTIONS)A.FILL IN THE BLANKS) |54 VideosCHEMICAL KINETICS
ICSE|Exercise ISC EXAMINATION QUESTIONS (NUMERICAL PROBLEMS )|57 VideosBIOMOLECULES
ICSE|Exercise ISC EXAMINATION QUSTIONS (PART-I)(DESCRIPTIVE QUESTIONS)|21 VideosCHEMISTRY IN EVERYDAY LIFE
ICSE|Exercise EXERCISE (PART - II (DESCRIPTIVE QUESTIONS) (LONG ANSWER QUESTIONS))|19 Videos
Similar Questions
Explore conceptually related problems
ICSE-CHEMICAL KINETICS-PROBLEM
- The kinetics of hydrolysis of methyl acetate in excess of hydrochloric...
Text Solution
|
- While studying the kinetics of the reaction involving conversion of am...
Text Solution
|
- For the reaction A to B +C, the following data were obtained fi...
Text Solution
|
- The optical rotation of sucrose in 0.90 N HCl at various times is give...
Text Solution
|
- The half-life period of a substance is 50 minutes at a certain concent...
Text Solution
|
- At a certain temperature, the half-life period for the decomposition f...
Text Solution
|
- Following data were obtained for the catalytic decomposition of ammoni...
Text Solution
|
- The rate constants of a reaction at 500 K and 700 K are 0.02 s^(-1) an...
Text Solution
|
- The first order rate constant for the decomposition of ethyl iodide by...
Text Solution
|
- The value of rate constant for a second order reaction is 6.7 xx 10^(-...
Text Solution
|
- Rate constant k of a reaction varies with temperature according to equ...
Text Solution
|
- If a first order reaction has activation energy of 25000 cal and a fre...
Text Solution
|
- If a first order reaction has activation energy of 25000 cal and a fre...
Text Solution
|
- The activation energy for a first order reaction is 60 kJ mol^(-1) in...
Text Solution
|
- For a reaction, the energy of activation is zero. What is the value of...
Text Solution
|
- The rate constant for the decomposition of hydrocarbons is 2.418 × 10^...
Text Solution
|
- The decomposition of a hydrocarbon follows the equationk = (4.5 xx 10 ...
Text Solution
|
- The rate of a reaction triples when temperature changes from 50^@ C to...
Text Solution
|
- A certain reaction is 50% complete in 20 minutes at 300 K and the same...
Text Solution
|
- The rate of a reaction increases four times when the temperature chang...
Text Solution
|