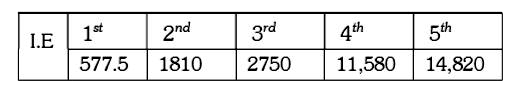
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
CLASSFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES
ERRORLESS|Exercise NCERT BASED QUESTIONS (Electron Affinity)|21 VideosCLASSFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES
ERRORLESS|Exercise NCERT BASED QUESTIONS (Electronegativity)|18 VideosCLASSFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES
ERRORLESS|Exercise NCERT BASED QUESTIONS (Atomic and Ionic Radii)|39 VideosCHEMICAL EQUILIBRIUM
ERRORLESS|Exercise ASSERTION & REASON |12 VideosENVIRONMENTAL CHEMISTRY
ERRORLESS|Exercise ASSERTION & REASON|7 Videos
Similar Questions
Explore conceptually related problems
ERRORLESS-CLASSFICATION OF ELEMENTS AND PERIODICITY IN PROPERTIES -NCERT BASED QUESTIONS (Ionisation Energy)
- Which element having the following electronic configuration has minimu...
Text Solution
|
- The correct order of the second ionisation potential of carbon, nitrog...
Text Solution
|
- The correct match of contents in Column I with those in Column II is
Text Solution
|
- One electron species having ionization enegry of 54.4 eV is
Text Solution
|
- Amongst the following elements whose electronic configuration are ...
Text Solution
|
- The incorrect statement among the following is
Text Solution
|
- The first ionization energy of oxygen is less than that of nitrogen. W...
Text Solution
|
- Which of the following explanation is best for not placing hydrogen in...
Text Solution
|
- Which of the following order is wrong-
Text Solution
|
- Among the following transition elements, pick out the element/elements...
Text Solution
|
- The decreasing order of the first ionization energy (in kJ mol^(-1)) o...
Text Solution
|
- Order of first ionisation potentials of elements Li,Be,B, Na is
Text Solution
|
- The correct decreasing order of first ionisation enthalpies of five el...
Text Solution
|
- For one of the element various successive ionization enthalpies (in 1 ...
Text Solution
|
- Which is the wrong order for the stated property ? .
Text Solution
|
- The elements which occupy the peaks of ionisation energy curve are
Text Solution
|
- The first (Delta(i)H(1)) and second (Delta(i)H(2)) ionization enthalpi...
Text Solution
|
- Among halogens, the correct order of amount of energy released in elec...
Text Solution
|
- The first ionization enthalpies for three elements are 1214, 1680 and ...
Text Solution
|
- The electronic configuration of an element with the largest difference...
Text Solution
|