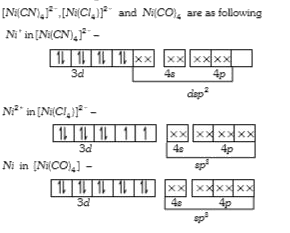
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
COORDINATION COMPOUNDS
ERRORLESS|Exercise NCERT BASED QUESTION (CRYSTAL FIELD THEORY)|17 VideosCOORDINATION COMPOUNDS
ERRORLESS|Exercise NCERT BASED QUESTION (COMPLEX STABILITY, SPECTROCHEMICAL SERIES AND EAN)|22 VideosCOORDINATION COMPOUNDS
ERRORLESS|Exercise NCERT BASED QUESTION (WERNER.S COORDINATION THEORY)|16 VideosCHEMISTRY IN EVERYDAY LIFE
ERRORLESS|Exercise Assertion & Reason|4 VideosELECTROCHEMISTRY
ERRORLESS|Exercise Assertion & Reason|11 Videos
Similar Questions
Explore conceptually related problems
ERRORLESS-COORDINATION COMPOUNDS-NCERT BASED QUESTION (VALENCE BOND THEORY AND GEOMETRY AND MAGNETIC NATURE OF COORDINATION COMPOUNDS)
- In Fe(CO)(5), the Fe larr CO sigma bond results by the overlap between...
Text Solution
|
- The number of unpaired electrons in Ni (CO)(4) is
Text Solution
|
- Which complex compound obeys 18-electron rule
Text Solution
|
- Both [Ni(CO)4] and [Ni (CN)4]^(2-) are diamagnetic The hybridisation...
Text Solution
|
- Which of the following complex is an outer orbital complex?
Text Solution
|
- Which one of the following is an outer orbital complex and exhibits pa...
Text Solution
|
- The diamagnetic species is
Text Solution
|
- The bonds in K4[Fe(CN)6] are
Text Solution
|
- Amongst Ni(CO)(4),[Ni(CN)(4)]^(2-) and NiCl(4)^(2-):
Text Solution
|
- The geometry of Ni(CO)4 and Ni(PPH3)2Cl2 are
Text Solution
|
- The species having tetrahedral shape is
Text Solution
|
- The hybridized state of Al^(3+) in the complex ion formed when AlCl3 ...
Text Solution
|
- Consider the following two complex ions : [CoF(6)]^(3-) and [Co(C(2)O(...
Text Solution
|
- Choose the correct statement
Text Solution
|
- In the complex [SbF(5)]^(2-), sp^(3) d hybridization is present. Geome...
Text Solution
|
- in [Cu(NH(3))(4)]SO4,Cu has following hybridization
Text Solution
|
- The spin-only magnetic moments of [Mn(CN)(6)]^(4-) and [MnBr(4)]^(2-) ...
Text Solution
|
- For a tetrahedral complex [MCl(4)]^(2-), the spin-only magnetic moment...
Text Solution
|
- The spin-only magnetic moments of [Fe(NH3)6]^(3+) and [FeF6]^(3-) in ...
Text Solution
|
- The spin only magnetic of [ZCl(4)]^(2-) is 3.87 BM where Z is
Text Solution
|