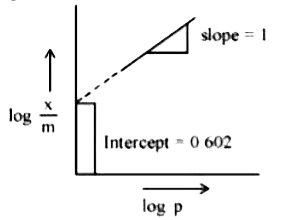
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
JEE MAINS PREVIOUS YEAR-JEE MAINS 2022-CHEMISTRY (SECTION-B)
- How many g atoms and number of atoms are there in (a) 60 g carbon, (b)...
Text Solution
|
- Consider the following set of quantum numbers . The number of ...
Text Solution
|
- BeO reacts with HF in presence of ammonia to give (A) which on therma...
Text Solution
|
- When 5 moles of He gas expand isothermally and reversibly at 300 K fro...
Text Solution
|
- A solution containing 2.5 xx 10^(-3) kg of a solute dissolved in 75xx ...
Text Solution
|
- pH value of 0.001 M NaOH solution is
Text Solution
|
- For the reaction taking place in the cell : Pt(s) H2 (g) H^+ (aq) ...
Text Solution
|
- It has been found for a chemical reaction with rise in temperature by ...
Text Solution
|
- If the initial pressure of a gass is 0.03 atm, the mass of the gas ad...
Text Solution
|
- 0.25 g of an organic compount containing chlorine gave 0.40 g of silve...
Text Solution
|