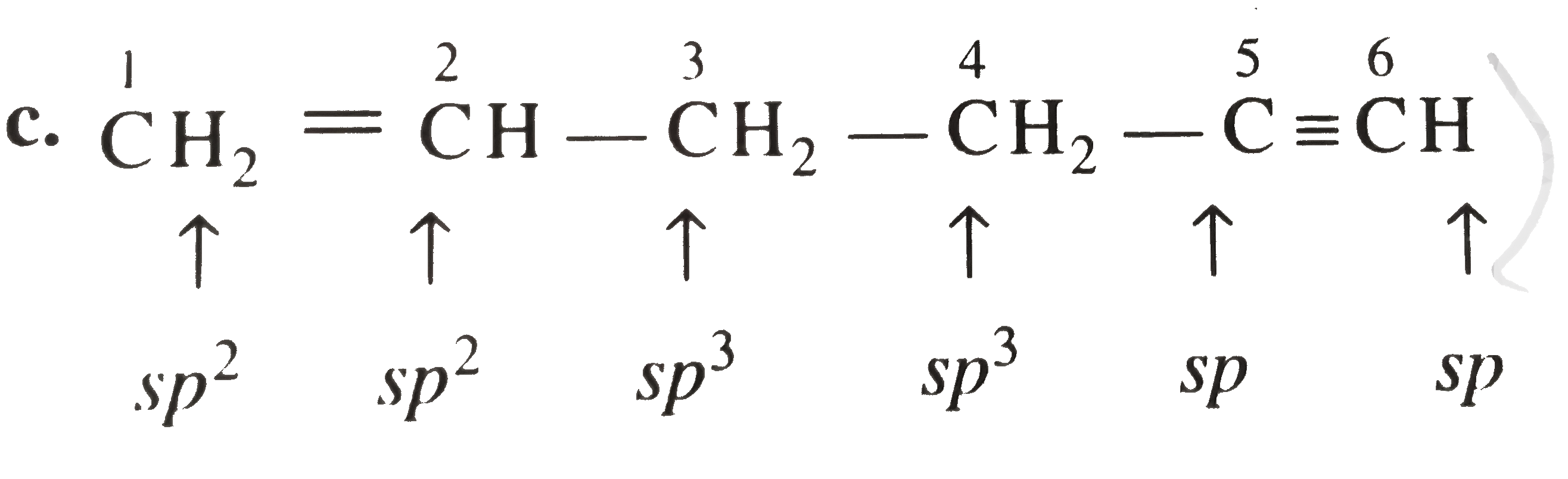A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
PURIFICATION OF ORGANIC COMPOUNDS AND QUALITATIVE AND QUANTITATIVE ANALYSIS
CENGAGE CHEMISTRY|Exercise Linked Comprehension Type|25 VideosPURIFICATION OF ORGANIC COMPOUNDS AND QUALITATIVE AND QUANTITATIVE ANALYSIS
CENGAGE CHEMISTRY|Exercise Multiple Correct Answers Type|10 VideosPURIFICATION OF ORGANIC COMPOUNDS AND QUALITATIVE AND QUANTITATIVE ANALYSIS
CENGAGE CHEMISTRY|Exercise Subjective Type|11 VideosPERIODIC CLASSIFICATION OF ELEMENTS AND GENERAL INORGANIC CHEMISTRY
CENGAGE CHEMISTRY|Exercise Exercises (Archives )Subjective|4 VideosREDOX REACTIONS
CENGAGE CHEMISTRY|Exercise Archives (Integers)|1 Videos
Similar Questions
Explore conceptually related problems
CENGAGE CHEMISTRY-PURIFICATION OF ORGANIC COMPOUNDS AND QUALITATIVE AND QUANTITATIVE ANALYSIS-Concept Application Type
- Describe the method to separate two compounds with different solubilit...
Text Solution
|
- What is the different betweeen distillation distillation under reduced...
Text Solution
|
- Why is nitric acid added to sodium extract before adding silver nitrat...
Text Solution
|
- Explain the reason for the fusion of an organic compound with metallic...
Text Solution
|
- Name a suitable technique of the components from a mixture of calcium ...
Text Solution
|
- Explain why an organic liquid vaporises at a temperature below its boi...
Text Solution
|
- Will C Cl4 give white precipitate of AgCl on heating with nitrate? Giv...
Text Solution
|
- Why is solution of potassium hydroxide used to absorb carbon dioxide e...
Text Solution
|
- Why is it necessary to use acetic acid and not sulphuric acid for the ...
Text Solution
|
- An organic compound contains 69% carbon and 4.8% hydrogen, the remaind...
Text Solution
|
- A sample of 0.50 gm of an organic compound was treated according to Kj...
Text Solution
|
- 0.3080 gm of and organic chloro compound gave 0.5740 gm of siver chlor...
Text Solution
|
- In the estimation of sulphur by carius method, 0.468 gm of an organic ...
Text Solution
|
- In the organic compound CH3==CH-CH2-CH2--=CH, the pair of hydridised...
Text Solution
|
- In the Lassaigne's test for nitrogen in an organic compound, the Pruss...
Text Solution
|
- Which of the following carbocation is most stable?
Text Solution
|
- The best and latest technique for isolation, purification, and separti...
Text Solution
|
- the reaction: CH3CH2I+KOH(aq.)rarrCH3CH2OH+KI is classified as:
Text Solution
|