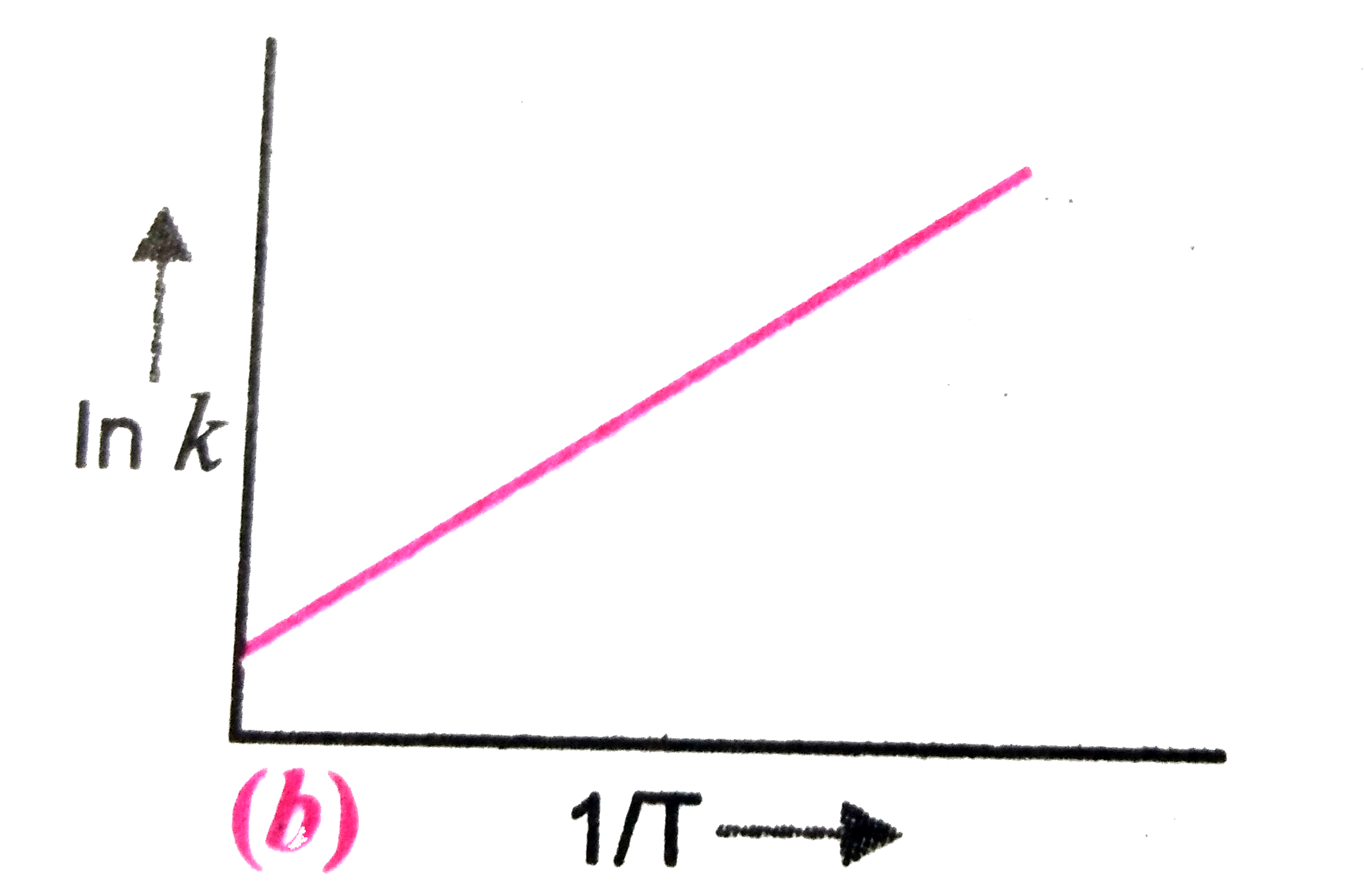
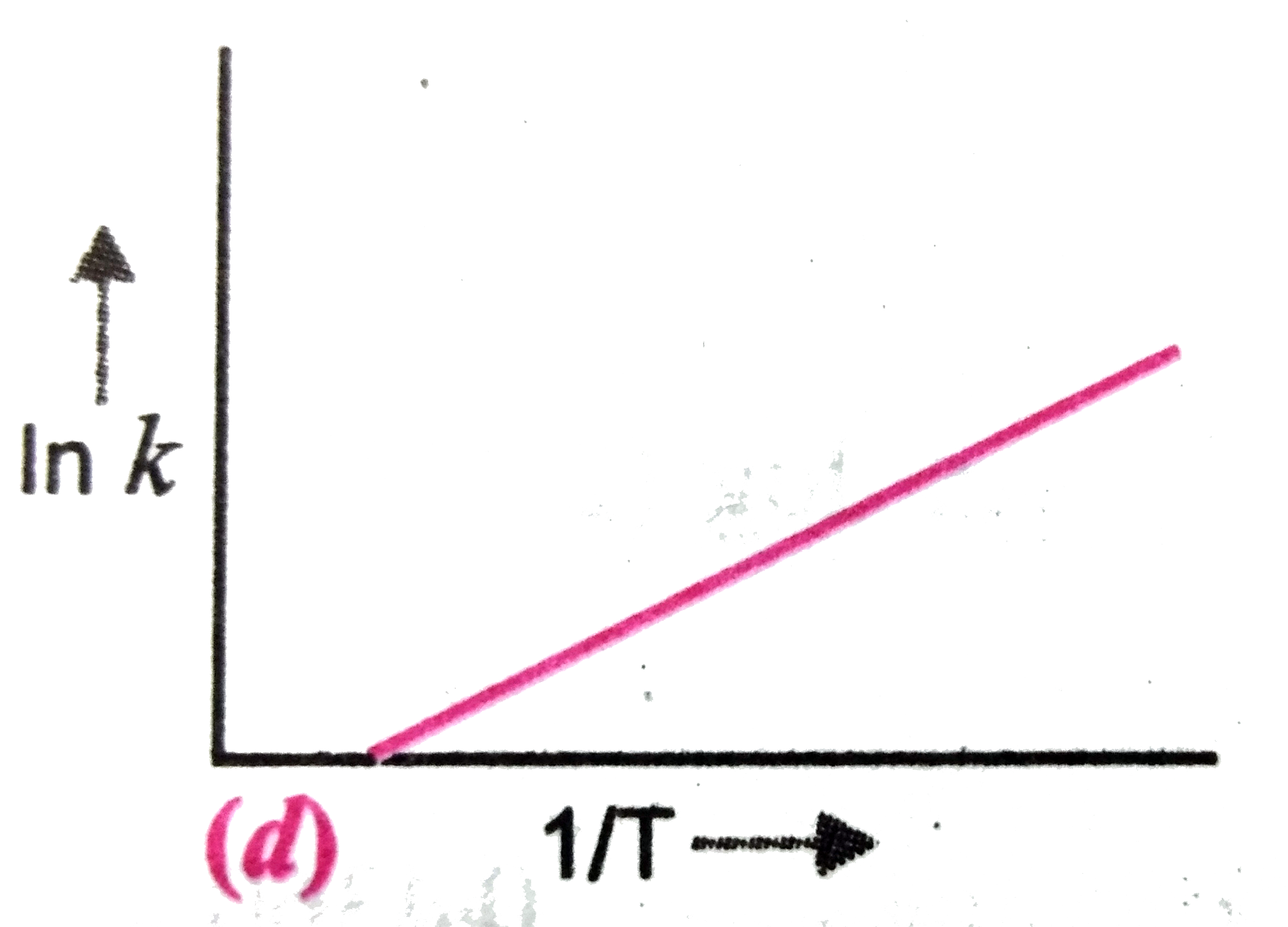
A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
CHEMICAL KINETICS
PRADEEP|Exercise NCERT EXEMPLAR PROBLEMS WITH ANSWERS, HINTS AND SOLUTTIONS (MULTIPLE CHOICE QUESTIONS-II)|12 VideosCHEMICAL KINETICS
PRADEEP|Exercise NCERT EXEMPLAR PROBLEMS WITH ANSWERS, HINTS AND SOLUTTIONS (SHORT ANSWER QUESTIONS)|20 VideosCHEMICAL KINETICS
PRADEEP|Exercise NCERT EXERCISES|30 VideosBIOMOLECULES
PRADEEP|Exercise IMPORTANT QUESTIONS (FOR BOARD EXAMINATION)|25 VideosCHEMISTRY IN EVERYDAY LIFE
PRADEEP|Exercise IMPORTANT QUESTION FOR BOARD EXAMINATION|30 Videos
Similar Questions
Explore conceptually related problems
PRADEEP-CHEMICAL KINETICS-NCERT EXEMPLAR PROBLEMS WITH ANSWERS, HINTS AND SOLUTTIONS (MULTIPLE CHOICE QUESTIONS-1)
- The role of a catalyst is to change
Text Solution
|
- In the presence of a catalyst, the heat evolved or absorbed during the...
Text Solution
|
- Activation energy of a chemical reaction can be determined by
Text Solution
|
- Consider Fig. below and mark the correct option.
Text Solution
|
- Consider a first order gas phase decomposition reaction given below: ...
Text Solution
|
- According to Arrhenius equation rate constant K is equal to A e.^(-...
Text Solution
|
- Consider the Arrhenius equation given below and mark the correct op...
Text Solution
|
- A graph of volume of hydrogen released vs time for the reaction betwee...
Text Solution
|
- Which of the following statement is not correct about order of a ...
Text Solution
|
- Consider the graph given in figure . Which of the following optio...
Text Solution
|
- Which of the following statements is correct ?
Text Solution
|
- Which of the following expression is correct for the rate of reac...
Text Solution
|
- Which of the following graphs represents exothermic reaction ?
Text Solution
|
- Rate law for the reaction, A + 2B to C is found to be Rate = k [A] [...
Text Solution
|
- Which of the following statements is incorrect about the collision the...
Text Solution
|
- A first order reaction is 50% completed in 1.26 xx 10^(14)s. How much ...
Text Solution
|
- Compounds 'A' and 'B' react according to the following chemical equati...
Text Solution
|
- Which of the following statement is not correct for the catalyst ...
Text Solution
|
- The value of rate constant of a pseudo first order reaction
Text Solution
|
- Consider the reaction A to B. The concentration of both the reactan...
Text Solution
|