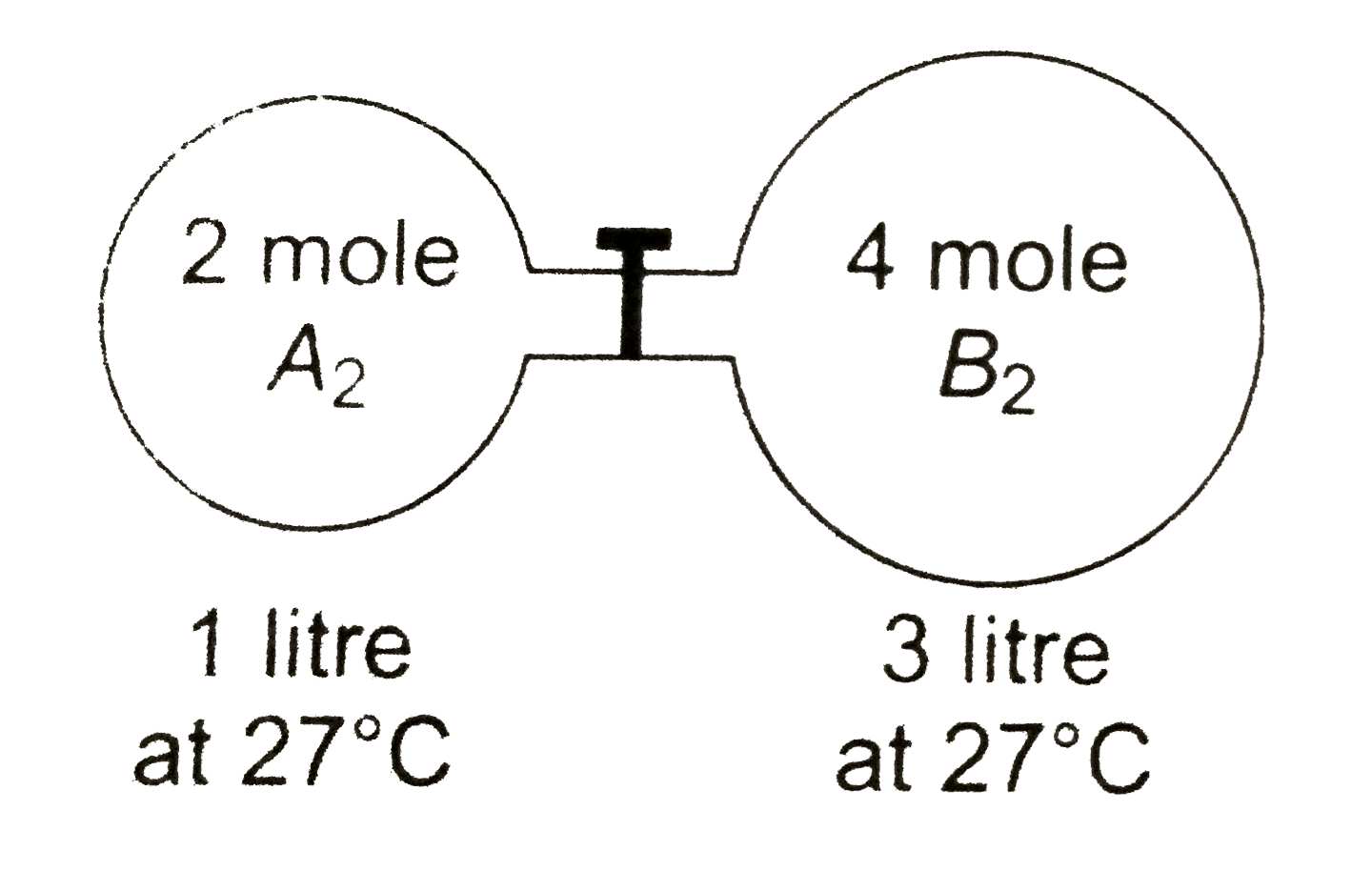A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
GRB PUBLICATION-CHEMICAL EQUILIBRIUM-D.Le Chateliera principle
- The gas A(2) in the left flask allowed to react with gas B(2) present ...
Text Solution
|
- For the reaction CO(g)+H2O(g) hArr CO2(g)+H2(g) at a given temperature...
Text Solution
|
- Given the following reaction at equilibium. N2(g) +3H2(g)hArr 2NH3(g...
Text Solution
|
- For an equilibrium H2O(s) hArr H2O(l)which of the following statements...
Text Solution
|
- When a bottle of cold drink is opened, the gas comes out with a fizzle...
Text Solution
|
- The equilibrium , SO2Cl2(g) hArr SO2(g)+Cl(2)(g) is attained at 25^@C ...
Text Solution
|
- An equilibrium mixture in a vessel of capacity 100 litre contains 1 mo...
Text Solution
|
- The following two reactions: i. PCl(5)(g) hArr PCl(3)(g)+Cl(2)(g) ...
Text Solution
|
- For the reaction, 4NH(3)(g) + 5O(2)(g)hArr4NO(g) + 6 H(2)O(l), Delt...
Text Solution
|
- For the reaction H(2)(g)+I(2)(g) hArr 2HI(g) The equilibrium const...
Text Solution
|
- To the system, LaCl(3)(s)+H(2)O(g) hArr LaClO(s)+2HCL(g)-"Heat" alre...
Text Solution
|
- In the Haber process for the industrial manufacturing of ammonia invol...
Text Solution
|
- For the reaction CaCO3(s)hArr CaO(s)+CO2(g) , the pressure of CO2 (g) ...
Text Solution
|
- Which of the following will not shift the equilibrium , N2+3H2 hArr 2N...
Text Solution
|
- One mole of helium (He) gas is added to the equilibrium N2O4(g) hArr 2...
Text Solution
|
- Some quantity of water is contained in a container as shown in figure....
Text Solution
|
- For the equilibrium CuSO(4)xx5H(2)O(s)hArrCuSO(4)xx3H(2)O(s) + 2H(2)O(...
Text Solution
|