Topper's Solved these Questions
Similar Questions
Explore conceptually related problems
SL ARORA-TRANSIENT CURRENT-Example
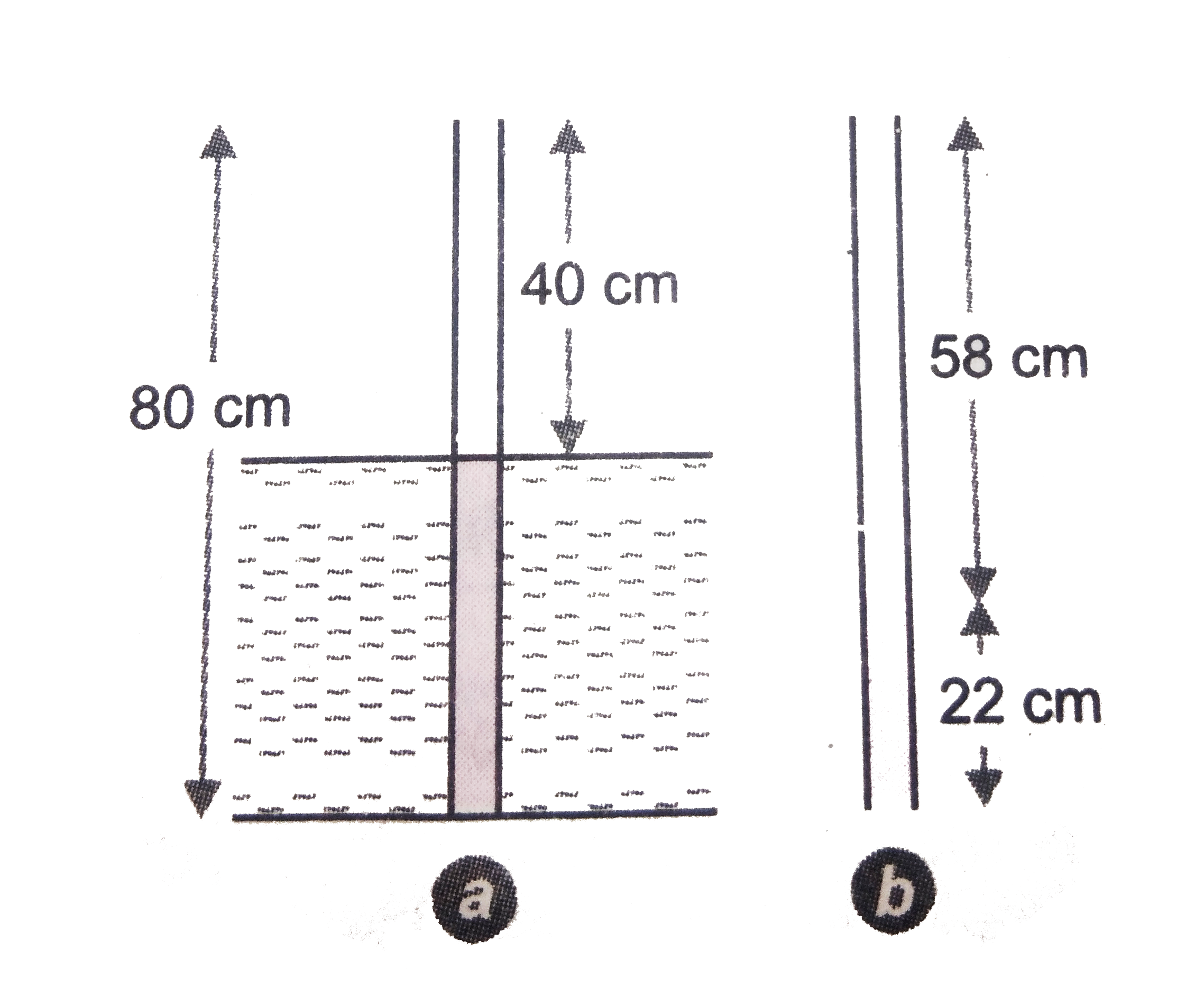
- A narrow uniform glass tube 80 cm long and open at both ends is half i...
Text Solution
|
- A metre long narrow bore held horizontally (and close at one end) cont...
Text Solution
|
- A given mass of a gas at -73^(@)C exerts a pressure of 50 cm of mercur...
Text Solution
|
- The rms velocity of hydrogen at S.T.P is u ms^(-1). If the gas is heat...
Text Solution
|
- A gas at 27^@ C in a cylinder has a volume of 4 litre and pressure 100...
Text Solution
|
- As an air bubble rises from the bottom of a lake to the surface, its v...
Text Solution
|
- Molar volume is the volume occupied by 1 mole of any (Ideal) gas at st...
Text Solution
|
- Using the ideal gas equation, determine the value of gas constant R. G...
Text Solution
|
- Molecular weight of oxygen is 32. At S.T.P., volume of 1 g of oxygen ...
Text Solution
|
- Estimate the total number of air molecules (inclusive of oxygen, nitro...
Text Solution
|
- An air bubble of volume 1.0 cm^(3) rises from the bottom of a lake 40 ...
Text Solution
|
- An oxygen cylinder of volume 30 litres has an initial gauge pressure o...
Text Solution
|
- A mixture of hydrogen and oxygen has volume 2000cm^(3), temperature 30...
Text Solution
|
- A vessel with open mouth contains air at 60^@C. When the vessel is hea...
Text Solution
|
- The density of nitrotgen is 1.25 kgm^(-3) at S.T.P. Find its density a...
Text Solution
|
- A vessel contains 14g of hydrogen and 96g of oxygen at STP. (a) Find ...
Text Solution
|
- A vessel contains two non-reactive gases neon (monoatomic) and oxygen ...
Text Solution
|
- Four molecules of a gas have speeds 2, 4, 6 and 8 kms^(-1) respectivel...
Text Solution
|
- If three gas molecules have velocity 0.5, 1 and 2 km//s respectively, ...
Text Solution
|
- Calculate the r.m.s. velocity of air molecules at S.T.P. Given density...
Text Solution
|