Text Solution
Verified by Experts
Topper's Solved these Questions
CLASSROOM PROBLEMS
MOTION|Exercise Radioactivity|23 VideosCLASSROOM PROBLEMS
MOTION|Exercise Solid State|18 VideosCLASSROOM PROBLEMS
MOTION|Exercise Chemical Kinetics|30 VideosChemical Kinetics
MOTION|Exercise Exercise - 4 (Level - II) (SUBJECTIVE PROBLEM)|1 VideosCLASSROOM PROBLEMS 1
MOTION|Exercise THERMODYNAMICS|17 Videos
Similar Questions
Explore conceptually related problems
MOTION-CLASSROOM PROBLEMS -Chemical Equilibrium
- The degree of dissociation of N2O4 in NO2 at 7 atm and 37^@C is 0.40. ...
Text Solution
|
- For the reaction 2NO(g) hArr N2(g) + O2(g) and NO(g) + 1//2Br2(g) hAr...
Text Solution
|
- In a gaseous reaction A+2B iff 2C+D the initial concentration of B was...
Text Solution
|
- For the reaction NOBr(g) hArr NO(g +1//2Br2(g), KP = 0.15 atm at 90^@...
Text Solution
|
- n mole of PCl(3) and n mole of Cl(2) are allowed to react at constant ...
Text Solution
|
- At temperature T, a compound AB2(g) dissociates according and the reac...
Text Solution
|
- Two gases A and B in the molar ratio 1 : 2 were admitted to an empty v...
Text Solution
|
- 16 moles of hydrogen and 4 moles of nitrogen are sealed in a one litre...
Text Solution
|
- The ratio of the rate of diffusion of a sample of N2O4 partially disso...
Text Solution
|
- N2O4(g) hArr 2NO2(g). In this reaction, NO2 is 20% of the total volu...
Text Solution
|
- For the reaction Ag(CN)(2)^(ɵ)hArr Ag^(o+)+2CN^(ɵ), the K(c ) at 25^...
Text Solution
|
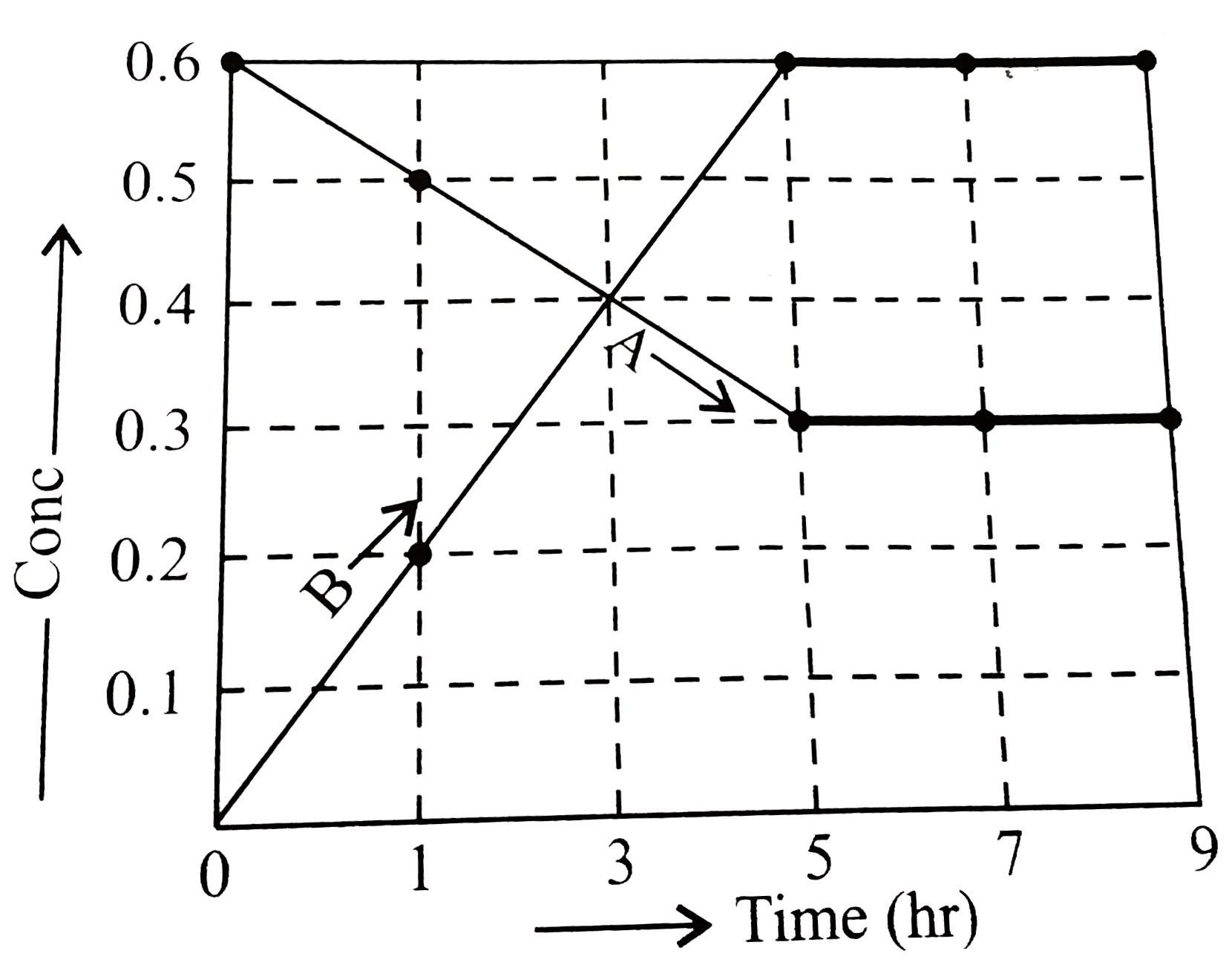
- The progress of the reaction A hArr nB, with times is presented in th...
Text Solution
|
- For NH(4)HS(s) hArr NH(3)(g)+H(2)S(g), the observed, pressure for reac...
Text Solution
|
- Some solid NH(4)HS is placed in flask containing 0.5 atm of NH(3). Wha...
Text Solution
|
- The equilibrium pressure of NH(4)CN(s) hArr NH(3)(g)+HCN(g) is 2.98 ...
Text Solution
|
- When 3.06 g of solid NH(4)HS is intoduced into a 2-L evacuated flask a...
Text Solution
|
- When 20 g of CaCO(3) were put into 10 litre flask and heated to 800^(@...
Text Solution
|
- Would 1% CO(2) in air be sufficient to prevent any loss in weight when...
Text Solution
|
- For the equilibrium: LiCl.3NH(3(s))hArrLiCl.NH(3(s))+2NH(3), K(p)=9 ...
Text Solution
|
- Two solid compounds A and B dissociate into gaseous products at 20^(@)...
Text Solution
|