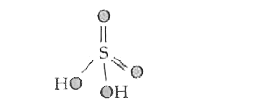Text Solution
Verified by Experts
Topper's Solved these Questions
P - BLOCK ELEMENTS
OSWAAL PUBLICATION|Exercise TOPIC - 3 (GROUP - 17 ELEMENTS, THEIR PROPERTIES AND SOME IMPORTANT COMPOUNDS)(VERY SHORT ANSWER TYPE QUESTIONS)|23 VideosP - BLOCK ELEMENTS
OSWAAL PUBLICATION|Exercise TOPIC - 3 (GROUP - 17 ELEMENTS, THEIR PROPERTIES AND SOME IMPORTANT COMPOUNDS)(SHORT ANSWER TYPE QUESTIONS)|11 VideosP - BLOCK ELEMENTS
OSWAAL PUBLICATION|Exercise TOPIC - 2 (GROUP - 16 ELEMENTS, THEIR PROPERTIES AND SOME IMPORTANT COMPOUNDS)(SHORT ANSWER TYPE QUESTIONS)|14 VideosORGANIC COMPOUNDS CONTAINING NITROGEN
OSWAAL PUBLICATION|Exercise CYANIDES , ISOCYANIDES AND DIAZONIUM SALTS PREPARATION , PHYSICAL AND CHEMICAL PROPERTIES USES ( Long Answer Type Questions-II )|3 VideosPOLYMERS
OSWAAL PUBLICATION|Exercise LONG ANSWER TYPE QUESTIONS - II|2 Videos
Similar Questions
Explore conceptually related problems
OSWAAL PUBLICATION-P - BLOCK ELEMENTS-TOPIC - 2 (GROUP - 16 ELEMENTS, THEIR PROPERTIES AND SOME IMPORTANT COMPOUNDS)(LONG ANSWER TYPE QUESTIONS - I)
- Complete the following equations : (i) 5SO(2)+2MnO(4)^(-)+2H(2)Orarr...
Text Solution
|
- Draw the flow chart for the manufacture of sulphuric acid by Contact p...
Text Solution
|
- (i) What happens when potassium chlorate is heated in presence of MnO(...
Text Solution
|
- Describe the preparation of Ozonised oxygen with an equation. Name the...
Text Solution
|
- Explain charring action of concentrated sulphuric acid on carbohydrate...
Text Solution
|
- Complete the following equations : (i) 2KClO(3)overset(MnO(2)" Heat"...
Text Solution
|
- Mention three anomalouos behaviour of oxygen.
Text Solution
|
- Complete the following equations i) CH(4)+2O(2)rarr ii) 2Fe^(3+)+S...
Text Solution
|
- Write the equation for i) The action of SO(2) with chlorine in the p...
Text Solution
|
- Write the equations involved in the manufacture of sulphuric acid in c...
Text Solution
|
- Justify the placement of O, S, Se, Te and Po in the same group of the ...
Text Solution
|
- Oxygen is a gas but sulphur a solid. Explain.
Text Solution
|
- Write balanced chemical equations for the following : (i) NaCl is he...
Text Solution
|
- Account for the following : (i) PCl(5) is more covalent than PCl(3)....
Text Solution
|
- Account for the following : (i) PCl(5) can act as an oxidising agent...
Text Solution
|
- How would you account for the following : (i) H(2)S is more acidic t...
Text Solution
|
- Account for the following : (i) Ammonia is more basic than phosphine...
Text Solution
|
- How would you account for the following : (i) The electron gain enth...
Text Solution
|