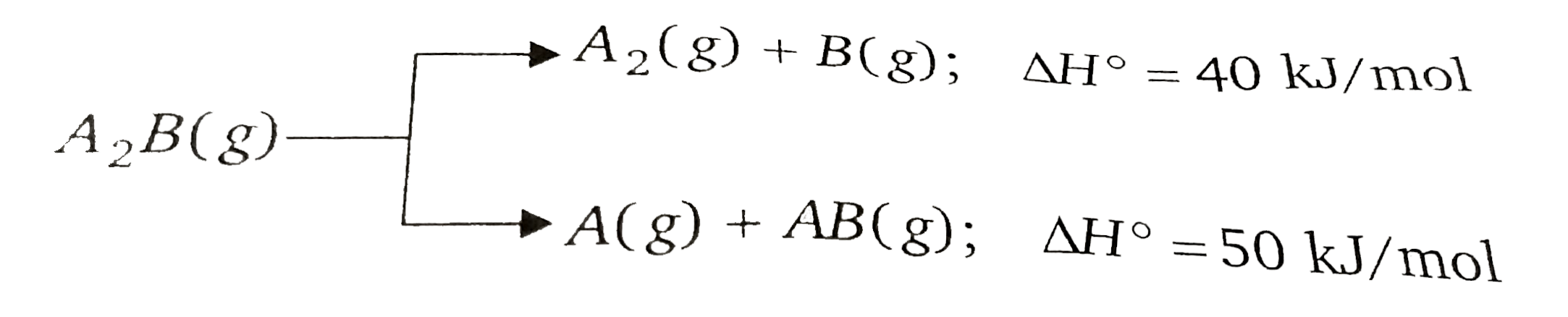Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
THERMODYNAMICS
RESONANCE ENGLISH|Exercise Exercise-2 Part-1: Only one option correct type|20 VideosTHERMODYNAMICS
RESONANCE ENGLISH|Exercise exercise-2 Part-2: Single and double value integer type|14 VideosTHERMODYNAMICS
RESONANCE ENGLISH|Exercise Main|5 VideosTEST SERIES
RESONANCE ENGLISH|Exercise CHEMISTRY|50 Videos
Similar Questions
Explore conceptually related problems
RESONANCE ENGLISH-THERMODYNAMICS-Exercise-1 Part-I Subjective question
- The heat of combustion of ethyl alcohol is -300 kcal. If the heats of ...
Text Solution
|
- Find out the heat evolved in combustion if 112 litres ( at STP) of wat...
Text Solution
|
- If {:(H(2)+1//2O(2)rarrH(2)O",",,,,DeltaH= -68 kcal),(K+H(2)OrarrKOH(a...
Text Solution
|
- Substance A(2)B(g) can undergoes decomposition to form two set of prod...
Text Solution
|
- One litre sample of a mixture of CH(4) and O(2) measured at 32^(@)C an...
Text Solution
|
- The standard enthalpy of decomposition of the yellow complex H(3)NSO(2...
Text Solution
|
- When 12.0g of carbon (graphite)reacted with oxygen to form CO and CO(2...
Text Solution
|
- Calculate the bond energy of Cl-Cl bond from the following data: CH(...
Text Solution
|
- Calculate DeltaH^(@) ("in" kJmol^(-1)) for the reaction CH(2)Cl(2)(...
Text Solution
|
- Calculate the enthalpy change (Delta H) of the following reaction 2C...
Text Solution
|
- Calculate change in enthalpy for the reaction at 27^(@)C H(2)(g)+Cl(...
Text Solution
|
- Estimate the average S-F bond enthalpy in SF(6). The values of standar...
Text Solution
|
- Calculate the standard enthalpy of solution of AgCl(s) in water DeltaH...
Text Solution
|
- Enthalpies of solution of BaCI(2)(s) and BaCI(2).2H(2)O(s) are -20.0kJ...
Text Solution
|
- The DeltaH(f)^(0)(KF,s) is -563 kJ mol^(-1). The ionization enthalpy ...
Text Solution
|
- Setup of Born-Haber cycle, calculation lattice energy of MgO((s)). The...
Text Solution
|
- Equal volumes of 1M HCI and 1M H(2)SO(4) are neutralised by 1M NaOH so...
Text Solution
|
- 150 mL of 0.5N HCl solution at 25^(@)C was mixed with 150 mL of 0.5 N ...
Text Solution
|
- The enthalpy of neutralization of a strong acid with a strong base is ...
Text Solution
|
- The standard enthalpy of neutralization of KOH with HCN and HCl in dil...
Text Solution
|