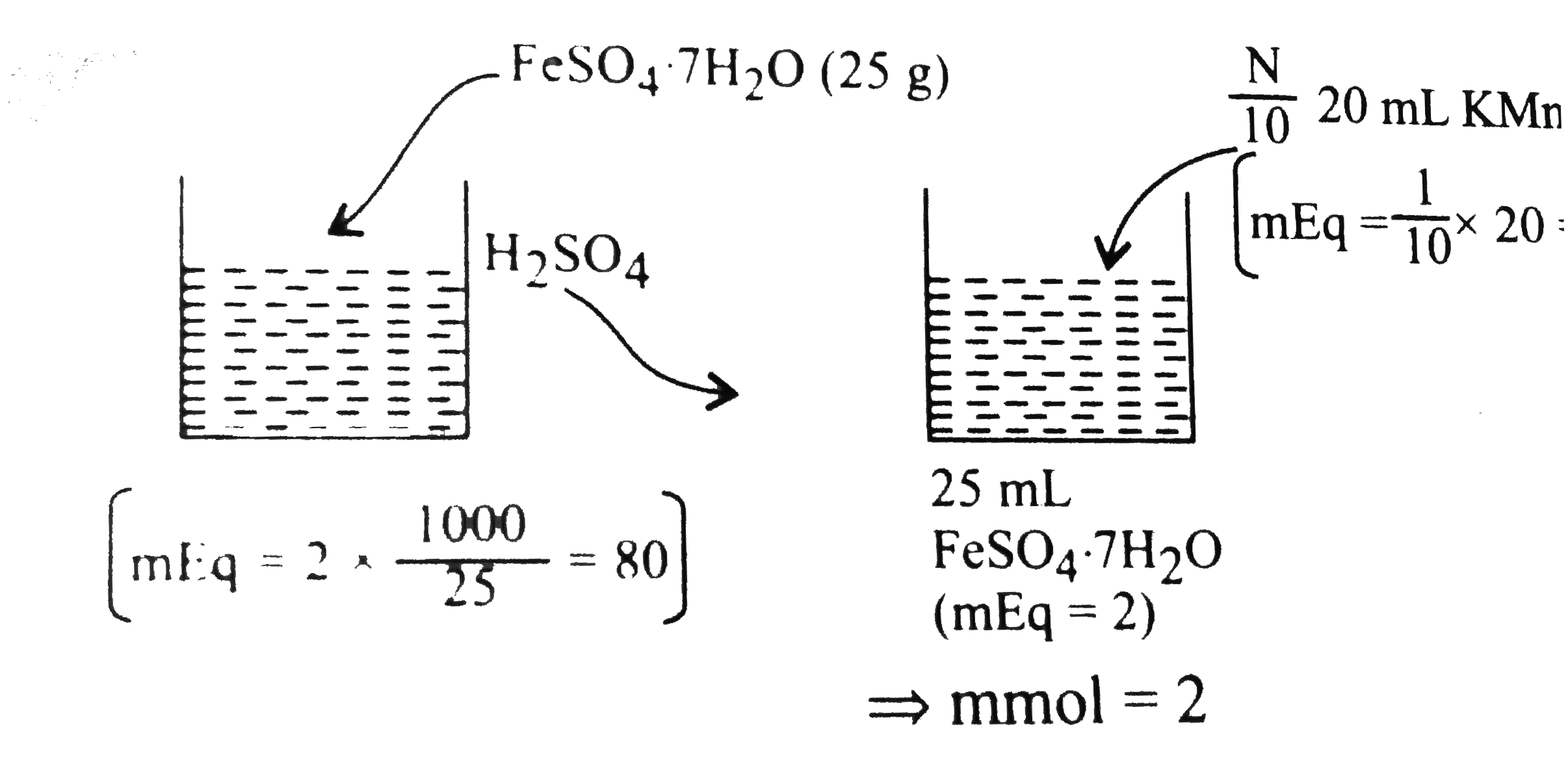A
B
C
D
Text Solution
Verified by Experts
The correct Answer is:
Topper's Solved these Questions
STOICHIOMETRY
CENGAGE CHEMISTRY|Exercise Exercises Assertion Reasoning|15 VideosSTOICHIOMETRY
CENGAGE CHEMISTRY|Exercise Exercises Integer|16 VideosSTOICHIOMETRY
CENGAGE CHEMISTRY|Exercise Exercises Multiple Correct|30 VideosSTATES OF MATTER
CENGAGE CHEMISTRY|Exercise Exercises (Ture False)|25 VideosTHERMODYNAMICS
CENGAGE CHEMISTRY|Exercise Archives (Subjective)|23 Videos
Similar Questions
Explore conceptually related problems
CENGAGE CHEMISTRY-STOICHIOMETRY-Exercises Single Correct
- 10mL of H2O2 solution (volume strength = x) requires 10mL of N//0.56 M...
Text Solution
|
- Equivalent weight of H(3)PO(2) when it disproportionates into PH(3) an...
Text Solution
|
- What volume of 0.1 M Ca(OH)(2) will be required neutralise 200 " mL of...
Text Solution
|
- What volume of 0.1 M Ca(OH)(2) will be required to neutralise 200 " mL...
Text Solution
|
- What volume of 0.2 M KOH will be requried to neutralise 100 " mL of " ...
Text Solution
|
- What volume of 0.1 M Ba(OH)(2) will be required to neutralise a mixtur...
Text Solution
|
- When 100 " mL of " 0.1 M Ba(OH)(2) is neutralised with a mixture of x ...
Text Solution
|
- If 10g of V(2)O(5) is dissolved in acid and is reduced to V^(2+) by zi...
Text Solution
|
- The volume of 0.5 M H(3)PI(4) that completely dissolved 3.1 g of coppe...
Text Solution
|
- 1 g of a sample of NaOH was dissolved in 50 " mL of " 0.33 M alkaline ...
Text Solution
|
- 0.4 g of polybasic acid HnA (all the hydrogens are acidic) requries 0....
Text Solution
|
- A mixutre solution of KOH and Na2CO3 requires 15 " mL of " (N)/(20) HC...
Text Solution
|
- 25.0 g of FeSO(4).7H(2)O was dissolved in water containing dilute H(2)...
Text Solution
|
- A 0.13 g of a specimen containing MnO(2) is treated with iodide ions. ...
Text Solution
|
- The mass of K(2)Cr(2)O(7) required to produce 5.0 L CO(2) at 77^(@)C a...
Text Solution
|
- A mixture of Na(2)C(2)O(4) and KHC(2)O(4).H(2)C(2)O(4) required equal ...
Text Solution
|
- The weight of MnO(2) and the volume of HCl of specific gravity 1.2 g m...
Text Solution
|
- How many moles of O(2) will be liberated by one mole of CrO(5) is the ...
Text Solution
|
- The purity of H2O2 in a given sample is 85%. Calculate the weight of i...
Text Solution
|
- 0.848 g aqueous solution of a mixture containing Na(2)CO(3) NaOH, and ...
Text Solution
|